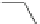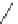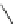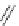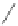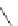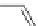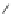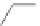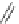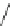Chemistry Reference
In-Depth Information
R
R
R
R
g
-Al
2
O
3
i
250 ºC
Cl
Cl
F
F
157a
(R = Me, 70%)
157b
(R = OMe, 91%)
157c
(R = H, 99%)
156c
156a
(R = Me)
156b
(R = OMe)
i: Pd(PCy
3
)
2
Cl
2
,DBU
DMAc, 140 ºC
g
-Al
2
O
3
250 ºC
98%
F
F
F
F
159
158
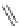
Scheme 49 Synthesis of
as
-indaceno[3,2,1,8,7,6-
pqrstuv
]picene derivatives [
168
,
169
]
and the bowl-to-bowl inversion barrier of buckybowls 151 (R-R
¼
C
2
H
4
) and 153
(R-R
¼
C
2
H
4
)[
164
] (Scheme
47
).
4.3 Circumtrindene
The FVP synthesis of circumtrindene (155) was conducted with decacyclene (154,
X
H) at 1,250
C, but this protocol gave the product in very low yield (Scheme
48
)
[
165
] because unfunctionalized aryl carbon atoms are inefficient for C-C bond
formation. Trichlorodecacyclene 154 (X
¼
Cl) dramatically improves the yield of
155 up to 27% [
166
]. Based on crystallographic analysis, the depth of the bowl is
3.107
¼
and the POAV pyramidalization angles of the central hexagon are 11.9
and 12.4
[
167
].
Å
5 Other Bulkybowls
5.1
as-Indaceno[3,2,1,8,7,6-pqrstuv]picenes
as
-Indaceno[3,2,1,8,7,6-
pqrstuv
]picenes 157a and 157b were efficiently synthe-
sized by Pd-catalyzed cyclization of dichlorobenzo[
s
]picenes 156a and 156b,
respectively (Scheme
49
)[
168
]. Recently, a new cyclization protocol through
regiospecific HF
elimination
on
fluoroarenes was
developed
[
169
].
-Al
2
O
3
at 250
C was efficiently converted to
indacenopicene 157c. One obvious advantage of this solvent-free protocol is
Difluorobenzo-[
s
]picene 156c in
ʳ