Environmental Engineering Reference
In-Depth Information
O
2
, 13% CO
2
, 80%, N
2
, 800 ppm NO, 1,200 ppm SO
2
, 600 ppm NO, 60 ppm HCl,
the results are displayed in Fig. 4.71.
10
Hg
0
(g)
Hg
2+
(g)
HgH
(g)
HgO
(g)
HgS
(g)
HgCl
(g)
HgCl
2(g)
HgSO
4(s)
T
o
tal H
g
8
Hg
HgCl
2(g)
6
4
HgSO
4(s)
2
HgO
(
g)
0
300
600
900
1200
1500
T
(K)
Fig. 4.71
Co-influence of typical gas components on mercury speciation
In general, during the pulverized coal combustion process, mercury in coal ex-
ists as Hg
0
in the flue gas in principle. The furnace temperature is around
12001500 °C, most of the mercury compound (organic or inorganic form) is in a
non-steady state, and tends to decompose into Hg
0
in a stable thermal state. With the
temperature falling gradually, the Hg
0
will be oxidized into Hg
2+
with the assistance
of oxidizing agents, chlorine, sulfur, iodine, oxygen, bromine, etc.
Conclusively, it was quite a little different from the previous result. As shown in
this figure, mercury started to exist in Hg
0
above 900 K, in particular. Within a
high-temperature region at about 1,500 K, mercury evaporated and Hg
0
was the
dominant form at that time. With the temperature cooling, mercury was oxidized
into HgO in a gaseous phase gradually, similar to the investigation result reported
that above temperature 1,073 K, mercury existed as Hg
0
primarily and HgO in a
very small amount
[30]
. At the same time, HgCl
2(g)
was converted into a big amount
swiftly, due to the fact that Cl
2
was rather more sensitive than O
2
. Accordingly, at a
temperature from 873 K to 1,273 K, gaseous Hg
0
, HgCl
2
and HgO co-exist at the
same time. At 873 K, HgCl
2
had completed 100% conversion. Almost all HgCl
2
conversion took place in this zone between 873 K and 373 K.
Up to 373 K, HgSO
4
in a solid phase would be formed and separated out from
the gas phase system. With the shift in chemical equilibrium, mercury will transfer
from HgCl
2
into HgSO
4(s)
.
Definitely, while in the low temperature region below 400 K, just as for ESP
and the WFGD tower entrance, mercury may exist in a gas-solid compound, i.e.
solid HgSO
4
, gaseous HgCl
2
, gaseous Hg
0
. As is known, HgCl
2
is highly soluble in
water, and small particles of HgSO
4
can be removed in the desulfurization tower.
Therefore, the utilization of the already existing WFGD tower for mercury removal
purposes is considered as a cost-effective, high-efficiency alternative.
The chemical thermodynamic equilibrium model is the ideal system response to
the results over an unlimited time. However, the residence time of the flue gas is
limited to only 3 5 s in the actual operation of the desulfurization tower. So the








































































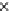





























































































Search WWH ::

Custom Search