Agriculture Reference
In-Depth Information
of ester production (Varma and Madras 2008), including chiral esters (Olsen
et al.
2006), kinetic resolution of secondary alcohols (Matsuda
et al.
2004a), lactones
(Bourne
et al.
2007) and enzyme catalysed oxidations (Amaral
et al.
2009) have
all been carried out in supercritical carbon dioxide. The ability to manipulate the
pressure and temperature of supercritical carbon dioxide has been shown to have
a signifi cant infl uence on the enantioselectivity of enzymes (Matsuda
et al.
2001,
2003) and this has been used to achieve high yields of homochiral alcohols.
The production of fl avour molecules using biocatalysts in supercritical CO
2
lends itself to a continuous process and this provides a cost-effi cient process,
which can be operated with relatively small process units. Continuous production
has been demonstrated for a number of product groups (Matsuda
et al.
2004b;
Laudani
et al.
2007). The equipment required is similar to that needed for
extraction with supercritical CO
2
( Fig. 12.7 ).
This relatively low temperature, mild process also enables more labile molecules
to be produced in higher yield than can be achieved using conventional routes. For
example, terpene esters can be produced in high yield by direct esterifi cation with
fatty acids (Olsen
et al.
2006; Peres
et al.
2003) that can be isolated easily from
other natural sources. The use of biocatalysis in supercritical fl uids is still in its
infancy, but small fl avour and fragrance molecules are ideal targets for this
technology and a greater range of affordable enzymes is becoming available.
12.3.2 Microwave production of fl avour bases
Conventional cooking with microwaves produces a complex series of reactions
which lead to characteristic food fl avours being produced. This same technology
Fig. 12.7
Typical components of a SCF reactor.
















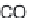




























Search WWH ::

Custom Search