Biology Reference
In-Depth Information
150,000
125,000
100,000
RLL
LRR
75,000
50,000
25,000
P
F1
BC1 BC2
BC3 BC4 BC5
BC6
Generations
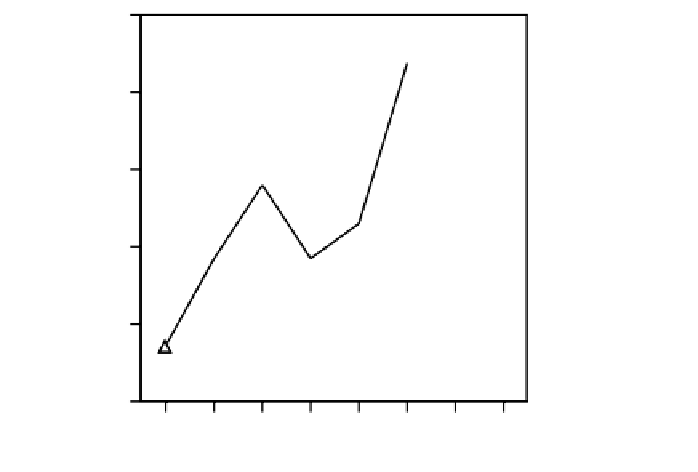
FIGURE 5.3
The
evolution of ovarian symbiont number in successive reciprocal backcrosses. LRR: intro-
duction of RR genome in LL cytoplasm, RLL: introduction of LL genome in RR cytoplasm. [ModiÝed from
Nardon, P., Grenier, A.M., and Heddi, A. (1998).
Symbiosis
25:
237Ï250.]
reciprocal F1 crosses and represents more or less the mean value of bacterial density in RR and
LL strains (Figure 5.3). Moreover, reciprocal backcrosses indicate that the symbiont population
decreases in LRR crosses when the RR genome is introduced into the LL cytoplasm, whereas it
increases in RLL lines where the LL genome is replacing the RR genome in the RR cytoplasm.
This demonstrates clearly that symbiont numbers are being controlled by a host genetic mechanism.
The molecular mechanism is currently under investigation using the cDNA subtraction approach,
which has revealed the existence of several bacteriocyte-speciÝc genes that may regulate bacterial
populations in symbiotic systems (A. Heddi et al.
,
unpublished data).
For
Wolbachia
endosymbioses, no extensive work has been carried out on this aspect, so it
remains unclear whether or not the host genetic system controls bacterial populations. However,
we know from previous work that not all weevil populations are infected with
Wolbachia
and not
all individuals are
Wolbachia
symbiotic when a given population is infected. Bourtzis
et al. (1996)
surveyed 41 stocks from the
Drosophila
genus and showed, with dot blot assays, that the infection
levels were not equal among populations. These results together provide support for bacterial control
by the host genetic system. However,
Wolbachia
genome variability could also explain why some
insect strains are infected while others are not at all or only partly infected.
W
OLBACHIA
I
NDUCE
C
YTOPLASMIC
I
NCOMPATIBILITY
IN
W
EEVILS
In insects,
Wolbachia
do not seem to improve the host
Ýtness
signiÝcantly except in one case
analyzed so far, where
Wolbachia
were shown to be necessary for the oogenesis of the parasitic
wasp
Asobara
tabida
(Dedeine
et al., 2001). Otherwise,
Wolbachia
are known to alter host
reproduction mainly in four ways: cytoplasmic incompatibility (Breeuwer and Werren, 1990;
OÔNeill and Karr, 1990), parthenogenesis (Stouthamer
et al., 1993), feminization of genetic males
(Rigaud
et al., 1991), and male killing (Jiggins
et al., 2000). In weevils, to investigate separately
and to distinguish the roles of SOPE and
Wolbachia
in host-symbiont biology, genetic analyses
were conducted with different crosses between individuals treated or not treated with heat or
tetracycline. Antibiotic treatment resulted in the elimination of both SOPE and
Wolbachia
, while