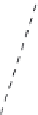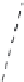Geoscience Reference
In-Depth Information
(a)
(b)
20,000
20,000
PVT
2 Molar K
2
HPO
4
PVT
2 Molar K
2
HPO
4
+0.125 Molar KNO
3
+KTP
18,000
84%
80%
18,000
84%
80%
75%
16,000
16,000
75%
14,000
E
14,000
G
12,000
12,000
D
10,000
10,000
8000
8000
C
6000
6000
4000
4000
F
2000
2000
H
A
B
0
150
0
150
200
250
300
350
400
450
500
200
250
300
350
400
450
500
Temperature (
°
C)
Temperature (
°
C)
Figure 5.37 PVT relations in KTP system
[148]
. (a) Pressure for various fills as a function
of temperature in 2 M K
2
HPO
4
. (b) Pressure for various fills as a function of temperature in
2MK
2
HPO
4
1
0.125 M KNO
3
saturated with KTP.
H
2
O
90
96
2m K
2
HPO
4
0.5-1.0 k Bar
325-425
°
C
94
D
92
90
XX
88
XXX
86
84
KTP +
KTP
KTP+TiO
2
(Anatase)
TiO
2
KPO
3
XXXX
82
A
EC
80
KTiOPO
4
Figure 5.38 Phase diagram for a KTP system
[148]
.