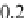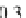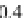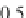Geoscience Reference
In-Depth Information
Fig. 1.16
Theoretical results for the total chemo-ionization rate constants in hydrogen
Tabl e 1. 6
Values of the
coefficients of PI and AI
reactions at
T
k
PI
10
10
k
AI
10
10
cm
3
s
1
cm
3
s
1
States
D
450 K
7
2
S
0.5
0.6
5
2
D
1.7
Negligible
6
2
D1.4
7
2
D1.2
8
2
D1.0
9
2
D0.9
A difference between the values
k
PI
and
k
AI
is defined by the difference of
contributions of the direct and the reverse processes to the total autoionization
width for
n
2
Sand
n
2
D states of the excited atom. Qualitatively, this can be seen
in the experiments on the hydrogen chemoionization of the alkali atoms and the
calculations for the hydrogen atom (Fig.
1.16
). A quantum mechanical model of
the chemoionization was considered by Duman and Shmatov (
1980
). The power
dependence
n
max
T
½
,where
n
max
is the value of the principal quantum number,
corresponds to the maximum of the rate constant.
1.8.2
Asymmetrical Collisions
The signals corresponding to the excitation states
n
2
P of the sodium atoms present
as minor impurities at optical excitation of the potassium vapor, as reported by