Geoscience Reference
In-Depth Information
Open system
Closed system
Amount of radiogenic
nuclide remaining
in the mineral
Temperature,
T
λ
P
≈
Closure temperature
T
c
Cooling age,
t
c
Time,
t
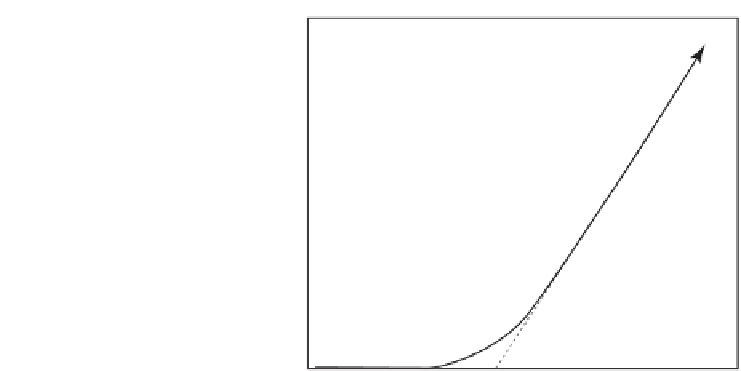
Figure 5.4
Cooling age and closure temperature of a chronometric system with decay constant
. The cooling
time and the corresponding cooling age
t
c
are defined by linearly extrapolating the ingrowth of
radiogenic nuclides at zero, while the closure temperature
T
c
is the temperature of the mineral at
this time. The system is open to the loss of the daughter isotope for
T>T
c
and closed for
T<T
c
.
The slope of the daughter-isotope evolution curve is simply the activity
λ
P
.
λ
Dividing
τ
c
by
τ
0
gives:
exp
E
i
RT
c
τ
c
τ
0
=
−
(5.15)
or
E
i
E
i
R
ln
D
0
θ/
T
c
=
(τ
0
/τ
c
)
=
τ
c
(5.16)
R
ln
a
2
We now express
θ
using
(5.13)
for
T
=
T
c
:
E
i
T
c
=
R
ln
D
0
RT
c
/
)
(5.17)
E
i
a
2
τ
c
(
−
d
T
/
d
t
which is Dodson's equation.
We can now evaluate
T
c
by trial and error and iteration. For example, let us consider the
closure of potassium feldspar crystals, assumed for simplicity to be spherical with a radius
of 2 mm, to the diffusion of
40
Ar produced by decay of
40
K. We assume that argon diffu-
sion is described by the following parameters:
D
0
=
10
−
6
m
2
s
−
1
,
E
i
1.4
×
=
180 000 J
mol
−
1
,
a
5KMy
−
1
. We would normally have to find the closure
temperature by trial and error and we would converge to
T
c
=
=
0.002 m, d
T
/
d
t
=
615 K. Upon inserting
342
◦
C
10
14
K, which corresponds to
T
=
=
θ
=
1.10
×
τ
0
=
T
c
.
We conclude that crystals of potassium feldspar with a radius smaller than 2 mm start los-
ing their radiogenic argon when heated above 342
◦
C, or, equivalently, that they start being
3.85
×
=