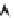Geoscience Reference
In-Depth Information
1
−
λ
230
Th
t
1
−
e
230
Th
232
Th
0
[
238
U
]
/
[
232
Th
]
Figure 4.10
The U-Th isochron. Samples derived from the same sedimentary or magmatic reservoir initially
have the same
230
Th
232
Th
isotope ratio but different U/Th ratios. They line up first on a
horizontal line and then migrate progressively toward the equiline, which is the stable state of
secular equilibrium. Square brackets indicate activity.
/
A first special application of the isochron method is that of the
230
Th dating method,
which is especially useful for recent volcanic rocks. If we re-write
(4.24)
by allowing for
the definition of excess
230
Th and if we then divide both sides by the activity of the thorium-
232 isotope (which decays slowly enough for this activity to be considered constant), we
obtain:
230
Th
232
Th
230
Th
232
Th
238
U
232
Th
1
e
−
λ
230
Th
t
e
−
λ
230Th
t
t
=
+
−
(4.29)
0
t
The right-hand side is the sum of two terms, which we will meet many times: the
first term expresses the demise of the initial condition, while the second term describes
how fast the system is moving towards steady state. It can be seen from an isochron
plot
x
with the same
230
Th
/
232
Th
0
ratio will lie on a horizontal line. With time the
alignment pivots around the point of intersection with the line
y
=
238
U
/
232
Th
,
y
x
, known as the equi-
line, until its slope becomes unity: at this point, the system is in secular equilibrium
230
Th
=
238
U
, see above
. The slope of the alignment gives an age, at least as long as
we are far from equilibrium. This method is often applied to minerals extracted from lavas
to date their crystallization. It can only be applied for ages of less than 350, 000 years.
A second special application is that of the lead-lead method. By utilizing the stable lead
isotope of mass 204, the two equations of the isochron can be written:
206
Pb
204
Pb
=
206
Pb
204
Pb
238
U
204
Pb
e
λ
238
U
t
1
t
−
0
=
−
(4.30)
t