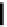Biomedical Engineering Reference
In-Depth Information
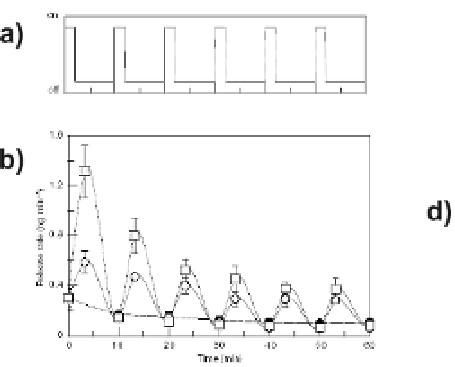
Fig. 4
The release rate of VEGF incorporated into alginate hydrogels is regulated by me-
chanical strain in vitro and cyclic strain results in enhanced angiogenesis in vivo [25].
Specifically, 6 cycles of mechanical stimulation were performed (mechanical compres-
sion for 2 min followed by relaxation for 8 min) and the VEGF release rate monitored in
vitro (
a
). Increased amplitude of strain led to increased VEGF release (10%=
open circles
,
25%=
open squares
, no compression =
filled circles
)(
b
). In vivo, mechanical stimulation
of alginate gels (
d
) yielded enhanced blood vessel densities relative to un-stimulated gels
(
c
).
Arrows
indicate blood vessels in the muscle tissue surrounding the implanted gels
(Nature [25], with permission of Nature Publishing Group)
189 and 206 may provide strong, localized signals [24]. Depending on the re-
spective clinical application, combined delivery of VEGF121 along with ECM
binding VEGF isoforms may prove beneficial by recreating the favorable func-
tions of both isoforms.
3.1.2
Modulation of Cellular Interactions
The appropriate receipt and transmission of VEGF signaling by cells is af-
fected by their interaction with the surrounding ECM and neighboring cells.
Hence, there is considerable interest in controlling the interfacial interactions
of polymeric delivery vehicles with the biological system in a way that mim-
ics natural processes. Integrins and cadherins are important families of cell
surface receptors that mediate cell-ECM and cell-cell interactions, respec-
tively [39, 40], and coordinately modulate VEGF signaling activity [41-43].
Integrins typically initiate cellular interactions with implanted polymeric de-
vices by recognizing and binding to adsorbed ECM molecules and communi-
cating through the cell membrane into the cells (outside-in signaling). Vice
versa, integrins alter cell-ECM interactions in response to intracellular sig-
nals (inside-out signaling) [40, 44]. This well-concerted interplay ultimately