Biomedical Engineering Reference
In-Depth Information
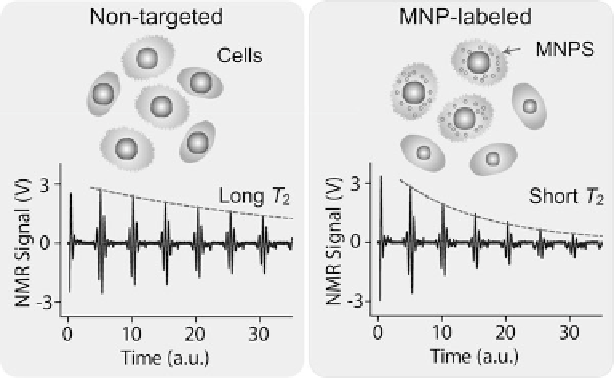
Fig. 9.1
Principle of DMR detection
. Biological objects (e.g., cells) tagged with MNPs can
accelerate the transverse relaxation of water protons. Compared to the nontagged samples, the
NMR signal will decay faster in time domain, therefore providing a sensing mechanism
of bioorthogonal targeting strategies [
27
,
29
] as well as accurate and real-time
control of device temperature [
23
], the DMR platform has become more robust
and sensitive, allowing operation in clinical settings [
26
]. This chapter reviews the
latest development of the DMR technology, focusing on its three major components:
magnetic nanoagents, miniature NMR systems, and optimized assay protocols.
Specific biomedical and clinical DMR applications will also be discussed.
9.2
Principle of DMR Detection
The DMR detection of MNP-labeled cells is realized by exploiting the
“T
2
-shortening” effect of MNPs in NMR measurements [
30
]. When placed in static,
polarizing magnetic fields for NMR detection, MNPs produce local dipole fields
with strong spatial dependence, which efficiently destroy the coherence in the spin-
spin relaxation of water protons. MNP-labeled objects consequently cause faster
decay of NMR signal, or shorter transverse relaxation time T
2
, than nontargeted
ones (Fig.
9.1
).
The capability of MNPs to induce T
2
changes is defined as transverse relaxivity
(r
2
)[
31
]. With MNPs in solution, the relaxation rate (R
2
D
1=T
2
) can be expressed
as [
28
]
N
P
V
;
R
2
D
R
W
C
r
2
(9.1)