Biomedical Engineering Reference
In-Depth Information
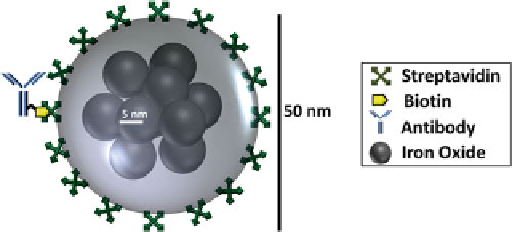
Fig. 7.4
Schematic representation of a magnetic nanoparticle (MNP) labeled with antibody drawn
to scale. The magnetic tag is comprised of a dozen iron oxide cores embedded in a dextran polymer
and then functionalized with antibody or receptor [
8
]
nanoparticle decreases, the magnetic moment decreases as well, causing the signal
per particle to decrease.
The solution to this problem is to cluster many MNPs into a dextran polymer
(Fig.
7.4
). In this way, the MNPs are physically isolated, allowing them to remain
superparamagnetic, while the overall magnetic content per magnetic tag is relatively
large due to the multiplicity of cores in each cluster.
7.3.2
Application of Magnetic Nanoparticles for Magnetic
Biosensing
Recent work has adapted magnetically responsive sensors for the detection of
biological species in solution by implementing a traditional sandwich assay directly
on these magnetically responsive nanosensors and utilizing superparamagnetic
nanoparticles as tags. If a magnetic particle similar to the one described above is
introduced to label the biomolecule of interest, magnetically responsive sensors are
capable of highly sensitive protein and oligonucleotide detection [
2
,
9
-
12
].
Among the more commonly used MNP tags in GMR biosensors are those com-
prised of clusters of monodisperse Fe
2
O
3
, superparamagnetic particles each with a
10 nm diameter embedded in a dextran polymer and functionalized with streptavidin
(Fig.
7.4
), as determined by TEM analysis [
13
]. The entire nanoparticle averages
46
13 nm in diameter (measured by number-weighted dynamic light scattering).
Based on the Stokes-Einstein relation, these particles have a translational diffusion
coefficient of approximately 8:56
˙
10
12
m
2
s
1
. The MNPs have a reported zeta
potential of
11 mV [
14
]. These particles are superparamagnetic and colloidally
stable, so they do not aggregate or precipitate during the reaction. Therefore, a
major advantage of using these tags is that the magnetically responsive sensors
detect the exact same signal before and after washing (Fig.
7.5
). This means that it
is equally valid to read the sensor signal prior to a final wash that removes unreacted