Biology Reference
In-Depth Information
Application of FRET and Microscopy
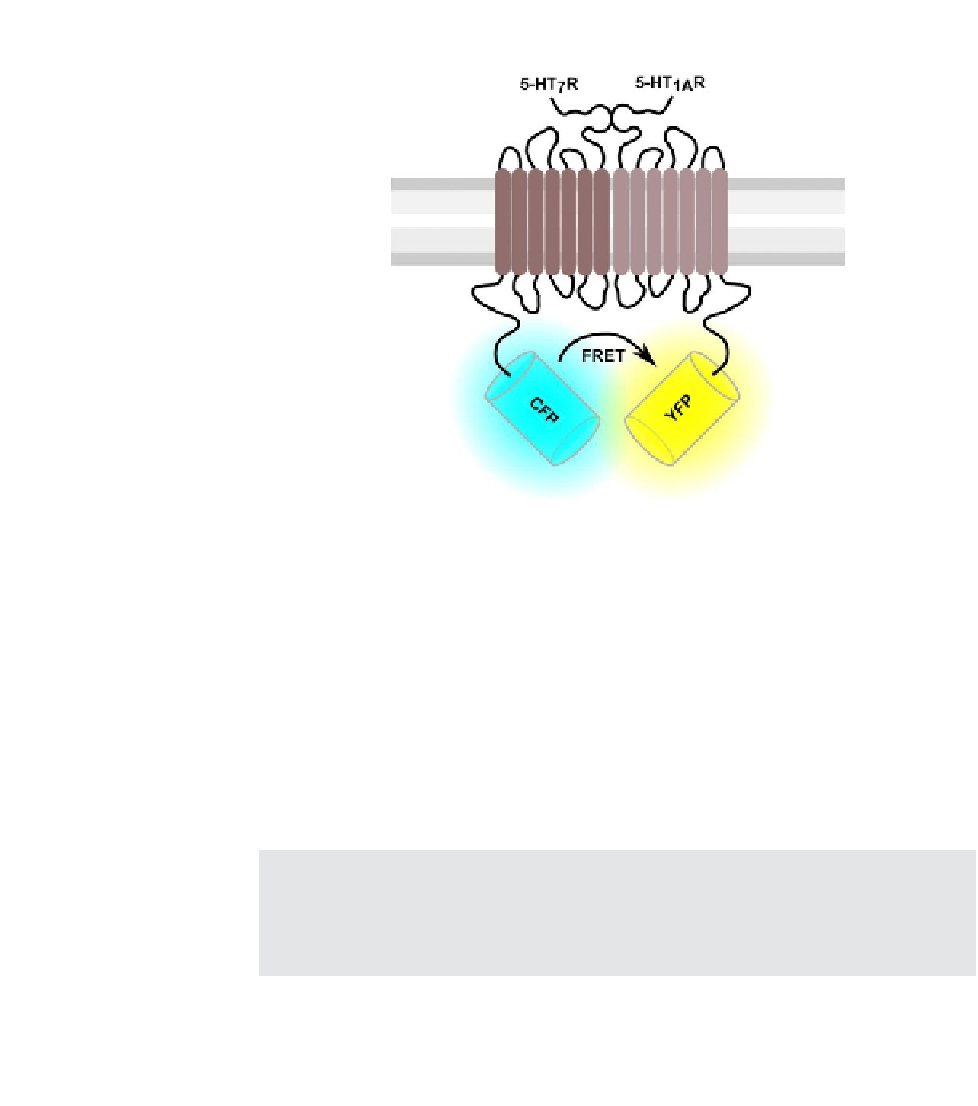
FIGURE 14.1
Heterodimerization of the serotonin receptors 5-HT
1A
and 5-HT
7
. The cartoon shows
intermolecular interaction between two proteins, one tagged with a cyan FP (donor) and the
other tagged with a yellow FP (acceptor). Only when specific interaction occurs (i.e., receptors
dimerizes), FRET signal can be measured.
state, which may transfer its excitation energy to a nearby acceptor fluorophore (or
chromophore) in a nonradiative fashion through long-range dipole-dipole interac-
tion. Basically, there are three major primary conditions required for FRET (see
Box and
Fig. 14.2
).
Primary conditions required for FRET are
close proximity of donor and acceptor molecules (typically 1-10 nm),
donor emission spectrumwith considerable spectral overlap acceptor excitation spectrum,
proper donor and acceptor transition dipole moment orientations.
The F¨rster theory shows that FRET efficiency (
E
) varies with an inverse sixth
power of the distance (
r
) between the two chromophores:
R
0
E
FRET
¼
(14.1)
r
6
þ
R
0
R
0
is the F¨rster distance where the FRET efficiency is 50% and typical values are
between 4 and 6 nm in case of acute FRET pairs. Therefore, it is suitable to employ
FRET for protein-protein interaction, because under physiological condition,