Biology Reference
In-Depth Information
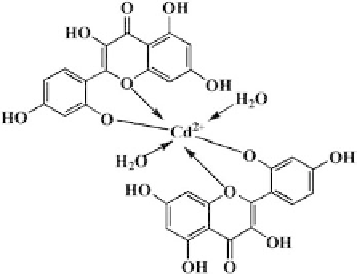
Scheme 7.2.
FormulaofCd(C
15
H
9
O
7
)
2
·
2H
2
O. Reprinted from Bioelectro-
chemistry, 73, S. Niu, M. Wu S., S. Bi, S. Zhang, Reaction of Cd(II)-Morin
with dsDNA for biosensing of ssDNA oligomers with complementary, GCE-
immobilized ssDNA, 64-69, 2008, withpermission from Elsevier.
Itsinteractionwithsalmonspermds-DNAwasinvestigatedusing
electrochemicalmethods.Thebindingstoichiometry(
m
=
1.76)and
10
−
5
Mwereevaluated
according to the Hill model for cooperative binding. Moreover,
Cd(morin)
2
wasusedasanindicatorthatallowedselectivedetection
of the target ss-DNA fragment. The target ss-DNA was quantified
over a linear range from 2.69
×
10
−
8
Mto9.16
×
10
−
7
M with a
detection limit of 9.30
×
10
−
9
M. In another report, interactions
of promethazine hydrochloride (PZH) with films prepared from
thiolated ss-DNA and ds-DNA on gold electrodes were studied by
Wei
et al.
[50]. The binding of PZH to the ss-DNA film is purely
based on an electrostatic interaction. However, the interaction of
the probe with the ds-DNA film is a combination of electrostatics
combined with intercalation into the duplex. The latter results in
an increased peak current for PZH oxidation and a larger electron
transfer coe
cient and afaster standard rate constant.
The use of [Cu(dmp)(H
2
O)]Cl
2
(dmp
=
2,9-dimethyl-1,10-
phenanthroline) as a new electrochemical hybridization indi-
cator was recently demonstrated by Li and coworkers [51].
[Cu(dmp)(H
2
O)]Cl
2
can intercalate into the base stack of ds-DNA
and has found applications for the detection of a Hepatitis B sensor
based on a synthetic21-mer ODN sequence.
=
.
×
equilibriumdissociationconstant
K
2
5