Biology Reference
In-Depth Information
Che and coworkers reported hexametallic platinum metallacycle
18
complexed with chelating dicarbene and cyanide ligands to
create a hexagonal cavity [71]. The platinacycle luminesces at 514
nm when subjected to MLCT (Pt
*
(carbene)) excitation in the
near-UV region. They observed that the emission was quenched by
N
−π
N
′
′
-bipyridinium hexafluorophosphate. However, the
metallacyclic supramolecules and their potential use as molecular
recognition probes remained unacknowledged until the report of
quantitative self-assembly of a Pd(II)-based molecular square
,
-dimethyl-4,4
19
by Fujita and coworkers in 1990 [72]. The X-ray structure of the
19
showed an almost perfect square with the pyridine rings slightly
twisted. The side-to-side distance in this square was approximately
8 Å. This molecular square showed the unique molecular recognition
ability of neutral aromatic guests such as benzene and naphthalene.
For instance, the square
encapsulates the electron-rich guest
1,3,5-trimethoxybenzene into the cavity in water (D
19
O) to form
2
−
1
a 1:1 host
H NMR chemical shift change
of 1.56 ppm for the aromatic protons and 0.59 ppm for methyl
protons in D
guest complex. The
O at room temperature were observed. Analysis of the
chemical shift change by Benesi
2
−
Hildebrand and a least-squares
procedure gave an association constant
−
1
C. Both
the hydrophobic and electrostatic interactions contribute to the
formation of host
K
= 750 M
at 25
°
a
−
guest complexes. Later they found that the square
19
showed smaller binding constant with smaller molecules such
−
as
p
-dimethoxybenzene (
K
= 330 M
1
),
m
-dimethoxybenzene (
K
=
a
a
−
1
−
1
580 M
), and
o
-dimethoxybenzene (
K
= 30 M
) [73].
a
8+
8NO
3
-
N
MeHN
NHMe
N
NH
2
N
NH
2
N
Pt
C
N
Pt
H
2
N
M
N
N
M
NH
2
MeHN
NHMe
N
N
N
C
MeHN
NHMe
C
N
N
N
18
N
Pt
t-Bu
N
N
N
N
MeHN
C
NHMe
H
2
N
M
N
N
M
NH
2
C
N
MeN
NH
2
NHMe
NH
2
Pt
N
C
Pt
N
N
M = Pd (
19
) and Pt (
20
)
N
N
NHMe
MeHN
OH
OH
OH
OH
OH
N
Me (PF
6
)
2
Guests
Me
N
Guest:
OH
OH
OH
HO
OH
OH
Stang
et al.
reported similar positively charged molecular squares
21
and
22
that recognize dihydroxynaphthalene in deuterated





















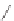





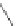
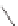

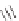



















































































































































































Search WWH ::

Custom Search