Geoscience Reference
In-Depth Information
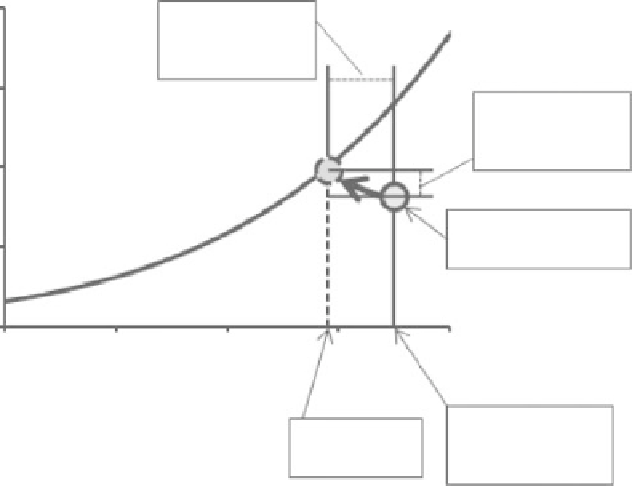
8
Air cools to
provide latent
heat
6
Air moistens
due to
evaporation
4
Vapor pressure
of the air (
e
)
2
0
Figure 2.4
Illustrating how
the air in a thermally
insolating container overlying
a thin layer of water saturates
and also cools to provide the
energy needed to evaporate
the water.
0
10
20
30
40
T
C
C
Air temperature
or “Dry bulb”
temperature
“Wet bulb”
temperature
Determining
T
wet
, the
wet bulb temperature
of a sample of air (see Fig. 2.3) while
also measuring air temperature is arguably the most common way that atmospheric
humidity is determined. In this context, the measured air temperature is generally
called the
dry bulb temperature, T
dry
. The names wet bulb and dry bulb temperature
are because air temperature can be measured using a mercury thermometer with
a dry 'bulb' (i.e., mercury reservoir), while a second mercury thermometer whose
bulb is moist is used to measure
T
wet
. In practice, the wet bulb is usually covered
with a moist cloth sheath that is shaded from the Sun's rays. Preferably both
thermometers should also be aspirated, i.e., have air drawn over their mercury
reservoirs, using a fan.
Wet bulb temperature is defined to be the temperature to which air is cooled
by evaporating water into it at constant pressure until it is saturated. It is help-
ful to consider the relationship between vapor pressure of air, and wet bulb and
dry bulb temperature by imagining a volume,
V
, of air overlying a thin layer of
water inside a container that thermally isolates the air from its surroundings.
Initially this air, which has a pressure
P
, has vapor pressure
e
and temperature
T
dry
. Some of the water in the thermally isolating container evaporates using
energy taken from the air itself to provide the required latent heat. Consequently,
the temperature of the air is progressively reduced. Eventually the air in the
container saturates. The now saturated air has the temperature
T
wet
and its
vapor pressure is equal to
e
sat
(
T
wet
), the saturated vapor pressure at this tem-
perature, see Fig. 2.4.
From Equation (2.9), it follows that the initial and final specific humidity of
the air in the container are
q
=
0.622(
e
/
P
) and
q
sat
(
T
wet
)
=
0.622[
e
sat
(
T
wet
) /
P
)],