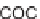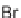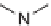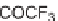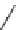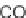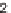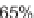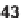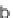Biomedical Engineering Reference
In-Depth Information
SCHEME 9.8
Folding pathway based on radical cyclization chemistry: (a) AIBN, Bu
3
SnH
or (Me
3
Si)
3
SiH, benzene, 80
◦
C; (b) KOH, MeOH-H
2
O; methoxyphenylisocyanate, Et
3
N,
THF, 45
◦
C.
complexity could be created when tandem radical cyclization processes were possible
(e.g., to yield
46
).
Oguri et al. developed a powerful approach in which metathesis was used to
“fold” alternative substrates into polycyclic ring systems inspired by artemisinin
(Figure 9.6) [26]. The folding substrates
51
to
56
were prepared in three or four steps
from the
-unsaturated nitrile
50
(Scheme 9.9). Subsequent folding using ring-
closing ene-yne-ene metathesis cascade chemistry generated the tricycles
57
to
62
.
Further synthetic transformations yielded analogs of the sesquiterpene artemisinin
(
63
to
65
), some of which exhibited antitrypanosomal activity.
,