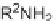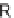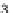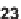Biomedical Engineering Reference
In-Depth Information
,
SCHEME 5.9
One-pot four-component synthesis of pyrrolo[3,4-
b
]pyridin-5-ones
23
.
TABLE 5.6 Pyrrolo[3,4-
b
]pyridin-5-ones 23
Entry
R
1
R
2
R
3
R
4
Yield (%)
a
1
n
-Hex
(CH
2
)
5
CO
2
Me
Bn
CO
2
Et
23i
,50
2
4-MeO-Ph
n
-Bu
Bn
CO
2
Et
23j
,55
3
n
-Hex
(
R
)-CH(PhCH
2
)CO
2
Me
Bn
CO
2
Et
23k
,54
4
n
-Hex
(
S
)-CH(PhCH
2
)CO
2
Me
Bn
CO
2
Et
23l
,48
5
n
-Hex
(
S
)-CH(CH
2
CO
2
Me)CO
2
Me
Bn
CO
2
Et
23m
,40
6
4-Cl-Ph
n
-Bu
Bn
4-NO
2
-Ph
23n
,32
7
n
-Hex
(CH
2
)
5
CO
2
Me
Bn
4-NO
2
-Ph
23o
,75
8
n
-Hex
n
-Bu
Bn
4-NO
2
-Ph
23p
,71
9
n
-Hex
2-[3
,4
-OMe)
2
]Ph(CH
2
)
2
Bn
4-NO
2
-Ph
23q
,70
10
Ph
n
-Bu
Bn
4-NO
2
-Ph
23r
,70
11
Ph
(CH
2
)
5
CO
2
Me
Bn
4-NO
2
-Ph
23s
,35
12
n
-Hex
(CH
2
)
5
CO
2
Me
Bn
4-OMe-Ph
23t
,65
a
Isolated yields.
SCHEME 5.10
Synthesis of tetracyclic pyrimidines.