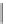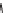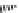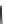Biomedical Engineering Reference
In-Depth Information
R
O
O
NO
2
N
R'
N
O
O
H
R'
•
cat. PPh
3
R
CH
2
Cl
2
or toluene
benzene, rt
O
O
O
O
O
O
39
40
41
Br
R'
EtO
2
C
=
R
,
OEt
,
CO
2
Et
OEt
,
,
CO
2
Et
SCHEME 4.13
Intramolecular [3
+
2] annulations and 1,3-dipolar cycloadditions.
broadening the scope of the electrophilic partner. When we used the electron-deficient
olefin
42a
as the electrophile, the [4
+
2] annulation of the
-methyl allenoate
7a
afforded functionalized cyclohexenes (Scheme 4.14) [50].
Notably, two distinct cyclohexene regioisomers were formed selectively, depend-
ing on the electronic nature of the phosphine catalyst. When we used hexam-
ethylphosphorus triamide (HMPT) as the catalyst, the reaction produced the cyclo-
hexene
44a
via the phosphonium dienolate intermediate
43
. On the other hand,
when we employed a more electron-withdrawing triarylphosphine, such as
tris
-(
p
-
fluorophenyl)phosphine, as the catalyst, the phosphonium dienolate
43
isomerized
into the vinylogous ylide intermediate
45
, leading to the reaction producing the
alternative cyclohexene regioisomer
46a
.
Under the optimized reaction conditions, both electron-deficient and electron-rich
arylidenes can be reacted with the
-methyl allenoate
7a
to provide the desired cyclo-
hexene products in high yields. Notably, these conditions also worked well for acti-
vated heteroarylidenes, furnishing the corresponding functionalized cyclohexenes.
The reaction tolerated a wide range of allenylic
-aryl substituents on
the allenoates, providing the cyclohexenes in excellent isolated yields with good to
excellent diastereoselectivities favoring the cis isomers (Scheme 4.15 and Table 4.5).
-alkyl and
Me
Me
NC
CN
CN
-
Ph
20 mol%
P(NMe
2
)
3
CO
2
Et + Ph
CO
2
Et
+
CN
P(NMe
2
)
3
7a
42a
CO
2
Et
benzene, reflux
43
44a
Ph
-
Me
NC
NC
CN
Me
20 mol%
CO
2
Et + Ph
CO
2
Et
+
CN
P(
p
-FC
6
H
4
)
3
P(
p
-FC
6
H
4
)
3
CO
2
Et
7a
42a
benzene, reflux
45
46a
SCHEME 4.14
Phosphine-catalyzed [4
+
2] annulations with arylidenemalononitriles.