Biomedical Engineering Reference
In-Depth Information
110
100
E
90
80
D
70
60
50
40
C
30
B
20
A
10
0
0
10
20 30
Time (minutes)
40
50
60
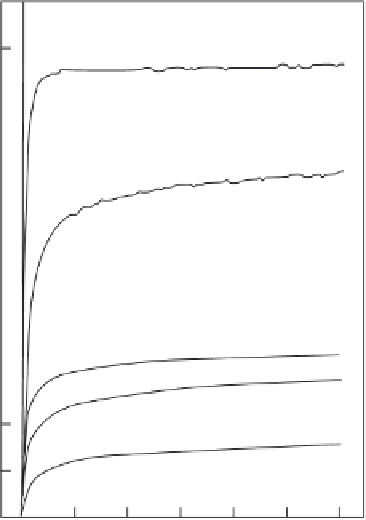
Figure 2.10.
Comparative dissolution of different calcium phosphates in acidic buffer. [64].
The calcium phosphates were prepared by precipitation. (A) Mg-substituted tricalcium phos-
phate,
β
-TCMP; (B) CHA; (C) Mg-substituted CHA; (D) OCP; (E) DCPD.
CaHPO
4
), occur in biologic systems (Table 2.1). Both DCPD and DCPA are used
in the preparation of apatites (calcium defi cient apatites, CDA) or substituted
apatites or substituted beta-tricalcium phosphate (e.g., Mg-TCP or Zn-TCP) in
the presence of the appropriate ions in solution during the hydrolysis of DCPD
or DCPA.
2.4.3 Octacalcium Phosphate (OCP)
OCP is structurally similar to apatite and has been speculated to be precursors to
biologic apatite [10]. OCP is formed by hydrolysis of DCPD or directly by pre-
cipitation, or in gel systems [61,64]. It can also be formed by electrochemical
deposition on metallic substrate [56,94]. OCP can transform to carbonate apatite
in the presence of
CO
2−
ions, to F-apatite in the presence of F
−
ions.
2.4.4 Tricalciumphosphate (
a
-TCP,
b
-TCP)
Pure beta tricalcium phosphate,
-TCP), cannot be obtained from
synthetic aqueous systems and it is therefore not surprising that it does not occur
β
- Ca
3
(PO
4
)
2
(
β






Search WWH ::

Custom Search