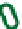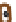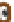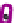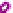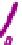Biology Reference
In-Depth Information
SC
GL
SL
BL
Figure 7.3
Hair follicle evagination in the Dicer cKO skin. In the Dicer-null skin,
developing hair follicles evaginate upward and arrest within the epidermis. Notably, the
epidermal differentiation is intact except in the region where the epidermal integrity is
disrupted by evaginating hair follicles. The same phenotype was also observed in the
Dgcr8-null skin, suggesting a causative role of the loss of stereotypical miRs for the
defects in hair morphogenesis.
These collective studies on
Dicer
cKO skin present the view that miRs in
the skin might be required to maintain the appropriate output of signaling
pathway(s) and, in turn, the finely tuned pathway(s) that are essential for the
maintenance of HF stem cells and the mesenchymal-epithelial cross talk that
orchestrates their proper downgrowth and lineage progression.
In striking contrast to hypoproliferation within the HF and the depletion
of its stem cells, hyperproliferation was observed in the mature
Dicer
cKO
epidermis (
Andl
et al
., 2006
). These defects suggest that the specific miRs
differentially expressed by these tissues may have functionally distinct roles.
If so, HF miRs would appear to control stem cell survival and maintenance,
while epidermal miRs seem more likely to govern cell cycle exit and/or the
balance between proliferation and differentiation.
Another differential feature of ablating
Dicer
in the skin was the preferen-
tial increase in apoptosis within the HFs, and particularly so within the highly
proliferative matrix compartment (
Andl
et al
., 2006; Yi
et al
., 2006
). The
specific localization of apoptotic cells was suggestive of either a general
requirement of Dicer (miRs) in rapidly dividing cells, consistent with apo-
ptotic phenotypes observed in Dicer cKO limb and T cells (
Harfe
et al
., 2005;
Muljo
et al
., 2005
), or a specific requirement for Dicer (miRs) in the hair
bulb. Recently, an exciting study has identified the
C. elegans
Dicer homolog
as a caspase substrate, and upon cleavage, the liberated Dicer RNase III
domain translocates to the nucleus and becomes a DNase that is critical
for the DNA fragmentation during apoptosis (
Nakagawa
et al
., 2010
). It is