Biomedical Engineering Reference
In-Depth Information
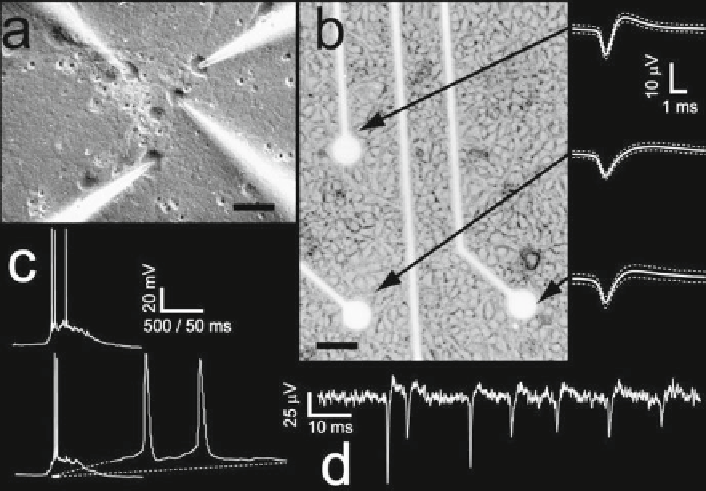
Fig. 5
Chronic detection of spontaneous electrical activity in neuronal cultures. Primary neuronal
cultures (
a-b
) can be maintained under healthy conditions for several weeks, growing on arrays of
substrate planar extracellular metallic electrodes (
b
). Extracellularly detected spikes (
b, d
) display
the stereotypical features of extracellular recordings. As apparent from the sample traces (
b, d
),
extracellular signals considerably differ from those obtained by means of simultaneous intracel-
lular multipatch recordings (
a
,
c
). Horizontal calibration: 25 mm in
a
, and 50 mm in
b
ing selectivity. In addition, there are key evidences indicating that CNT-based
materials display a peculiar signal coupling resembling an intracellular (i.e., patch)
and not extracellular access to the intracellular membrane potential (Liopo et al.
2006
; Mazzatenta et al.
2007
; see also Schoen and Fromherz
2007
) . Such a coupling
cannot occur at the interface between macroscopic metal electrodes and neuronal
membranes due to the generally smooth surface and lack of nanostructures. This is
shown in Fig.
6
, where a train of sustained action potential is evoked by CNT-
mediated electrical stimulation, reminiscent of a sustained direct intracellular cur-
rent fl ow. Although interpretation of these data requires careful discussion and an
assessment on the details of electrophysiological technique (see Mazzatenta et al.
2007
for a discussion), such evidences point out that intimate mechanical proximity
between bundles of CNTs and the neuronal membrane (Fig.
3f
) might correlate to
an intracellular-like access of the cytosolic cell compartments.
For the general character of our considerations, 2-dimensional morphologies of
a cultured neuron (Fig.
7a
), as reconstructed and digitized from microscope images
through a basic camera-lucida tracer (freely available at the Matlab Central Web
site, fi le id: 8336, The Mathworks, Natick, MA), can be reduced to a 1-dimension