Biomedical Engineering Reference
In-Depth Information
Period where
clinician sees effect
Period where healthcare
provider/society sees effect
Q
A
L
Y
competitor
your device
1
2
10
Years
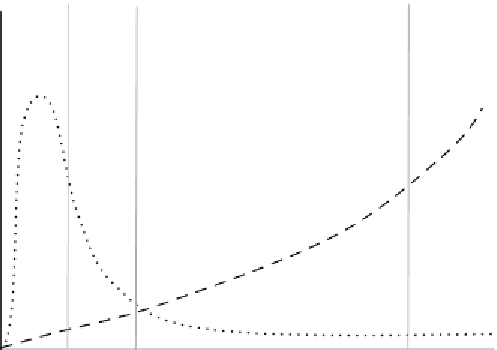
Figure 9.19
Example of a QALY graph demonstrating benefits over a long period.
If you find the development of
Table 9.8
daunting, try using the radial thinking technique (and
the other creativity techniques) illustrated earlier.
It is worth mentioning quality of life. Some healthcare providers use QALY as a measure
(Quality Adjusted Life Years - most now write QUALY). It is a very simple concept. It is a
measure that takes into account a small benefit to quality of life, but one which is enduring
as opposed to a quick fix remedy that has limited life. For example your device may have an
immediate requirement for antibiotics and painkillers, but this requirement lasts a few weeks.
This is compared with a competitor device that has no need for antibiotics but which has the
patient taking painkillers for the rest of their life. A QALY measure over the first year would
probably put your device well down the table of “good devices.” However, measuring QALY
over the whole lifespan of the patient puts your device out in front.
Figure 9.19
attempts to
illustrate this in graphical form. The graph also shows that portions of the graph have greater
influence on different end-users. For example a clinician may only see the patient during the
early treatment and then have a follow up after 12 months. Beyond that their interaction with
the patient may be zero. However, long-term costs for treatment of pain, etc., are borne by
society and the healthcare provider hence they are very interested in the aftereffects.
9.5.2 Stating Clinical Benefits
While you may have assessed your device's benefits and have some idea of what it can
achieve, until this is proven you are not allowed to state its benefits in any literature or
marketing material. Hence there is a need for an evaluation by a clinician. There is little doubt








Search WWH ::

Custom Search