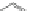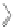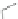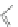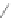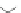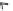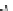Biomedical Engineering Reference
In-Depth Information
4.2
Basic Principles of “QD-Dye” Nanoassembly Formation
4.2.1
Characterization of Assembly Constituents
For ensemble and single particle experiments highly monodisperse colloidal
core/shell CdSe/ZnS QDs passivated by TOPO or long-chain amines (AM) as
well as uncapped CdSe QDs have been used (Evident Technologies, Inc, Troy, NY,
USA) (Fig.
4.1
). The main structural and optical properties of QDs are presented in
our earlier publications [
62
,
63
,
65
,
75
,
90
].
With respect to the formation of “QD-Dye” nanoassemblies, our focus is on
dye molecules of two classes:
meso
-pyridyl substituted porphyrins, (
m
-Pyr)
n
-H
2
P
or H
2
P (synthesis and properties are described in [
107
,
108
]) (Fig.
4.2
), and high
photostable pyridyl functionalized perylene diimide molecules, PDI [
109
,
110
]
(Fig.
4.3
).
CdSe Core
ZnS Shell
N
Key to the surface
TOPO
NH
2
AM
Evident Technologies, Inc.
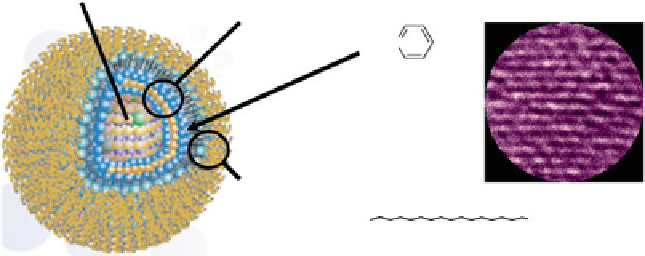
Fig. 4.1
Schematic structural presentation and transmission electron microscope image of
CdSe/ZnS QD. Tri-
n
-octyl phosphine oxide (TOPO) and long-chain amine (AM) ligands are also
shown. Key to the surface presents the basic scheme of functional anchor for a dye molecule
Porphyrin
abbreviation
Meso-substituents
r
m
= 0.75 nm
N
meta-pyridyl ring,
(m-Pyr) :
2
(
m
-Pyr)
1
-H
2
P
1: (
m
-Pyr)
2, 3, 4: (Ph)
para-pyridyl ring,
(p-Pyr) :
ortho-pyridyl ring,
(o-Pyr) :
N
NH
N
N
1
3
(
m
-Pyr)
2
-H
2
P
1, 3: (
m
-Pyr), opposite
2, 4: (Ph)
N
HN
phenyl ring, (Ph):
(
m
^Pyr)
2
-H
2
P
1, 2: (
m
^Pyr), adjacent
3,4: (Ph)
h = 1.0 nm
4
(
m
-Pyr)
3
-H
2
P
1, 2, 3: (
m
-Pyr)
4: (Ph)
Fig. 4.2
Chemical structure, abbreviations, and positions of pyridyl-substituents for
meso
-pyridyl
substituted porphyrins (H
2
P). The same scheme applies to para- or ortho pyridyl substituted H
2
P