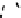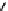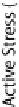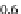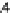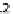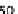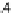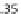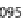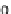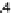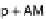Biomedical Engineering Reference
In-Depth Information
Fig. 4.3
Left
: fitting results with the model by Hai and Murphy (
1988
) where the active stress is
n
AMp
+
n
AM
and the myosin phosphorylation is
n
Mp
+
n
AMp
. The phosphorylating reaction rates
k
1
and
k
6
were set to 0
.
35 s
−
1
for 5 s followed by 0
.
085 s
−
1
. The other reaction rates were set to
0
.
1s
−
1
,
k
4
0
.
11 s
−
1
,
k
3
0
.
44 s
−
1
0
.
005 s
−
1
.
Right
: steady-state values
k
2
=
k
5
=
=
=
and
k
7
=
of the sum of fractions
n
AMp
+
n
AM
and
n
Mp
+
n
AMp
for different values of the phosphorylating
reaction rates
k
1
and
k
6
(Hai and Murphy,
1988
)
can be summarized by the following system of differential equations, i.e.
⎡
⎣
⎤
⎦
=
⎡
⎣
⎤
⎦
⎡
⎣
⎤
⎦
n
M
n
Mp
n
AMp
n
AM
−
k
1
k
2
0
k
7
n
M
n
Mp
n
AMp
n
AM
d
d
t
k
1
−
(k
2
+
k
3
)
k
4
0
,
0
k
3
−
(k
4
+
k
5
)
k
6
0
0
k
5
−
(k
6
+
k
7
)
(4.1)
where
n
M
,
n
Mp
,
n
AMp
and
n
AM
are fractions of the myosin functional states M, Mp,
AMp, AM, with the constraint
n
M
+
0 and
k
1
,...,k
7
are reaction rates describing the transition between the different functional states.
Hence, the reaction rates
k
1
and
k
6
represents the phosphorylation of M to Mp and
AM to AMp by the MLCK activity and the reaction rates
k
2
and
k
5
represents
the dephosphorylation of Mp to M and AMp to AM by the MLCP activity. The
reaction rates
k
3
and
k
4
represents the attachment and detachment of the cycling
phosphorylated cross-bridges and the reaction rates
k
7
represents the detachment
of the latch-bridges. The phosphorylating reaction rates
k
1
and
k
6
can be coupled
to the internal and also the external
n
Mp
+
n
AMp
+
n
AM
=
1,
n
i
≥
Ca
2
+
]
. Figure
4.3
shows the evolution of the
different fraction of the functional states with time using the model by Hai and
Murphy (
1988
).
When assuming maximal stimulated activation the phosphorylating MLCK ac-
tivity can be related and coupled to the extracellular
[
Ca
2
+
]
. In Murtada et al.
(
2010a
) a Michaelis-Menten kinetics characteristic of the MLCK activity was im-
plemented. The rate constants
k
1
and
k
6
are expressed as
[
2
α
Ca
2
+
e
,
[
CaCaM
]
k
1
=
k
6
=
,
[
CaCaM
]=
(4.2)
K
CaCaM
2
[
CaCaM
]
+