Biomedical Engineering Reference
In-Depth Information
to immobilize peptides on the pendant amino groups
onto the lysine side chains, while carboxyl-functionalized
PLA was prepared by copolymerizing side-chain-
protected b-alkyl-a-malate and subsequent deprotection
of the carboxyl groups by hydrogenation or alkaline
treatment.
the resulting polymer chains become again water soluble,
being absorbed in the body, followed by excretion from
kidney or bile duct, depending on the molecular size.
Naturally derived polymers including collagen, gelatin,
fibrinogen, albumin, polypeptides, HAc, CS, agarose,
alginate, and chitosan can also provide hydrogels.
Poly(ethylene glycol) or poly(ethylene oxide) (PEO)
with the same chemical structure as PEG is one of the
synthetic polymers that is most commonly used for
hydrogel fabrication in tissue engineering and is currently
approved by FDA for several applications. Each end of
PEG chains can be modified with either acrylates or
methacrylates to facilitate photocrosslinking. When the
modified PEG is mixed with an appropriate photo-
initiator and crosslinked via UV exposure hydrogels are
formed. This photo-induced method to produce hydro-
gels facilitates diverse, minimally invasive applications
via arthroscopy/endoscopy or subcutaneous injection
for tissue replacement or augmentation. Thermally re-
versible hydrogels have also been created from block
copolymers of PEG and PLLA.
Although
in situ
forming hydrogels offer advantages as
cell delivery carriers, cells do not usually adhere to highly
hydrated gels because of no built-in cell-adhesive ligands
and limited protein adsorption, so that the cells encap-
sulated inside gels are present in a ''blank'' environment
wherein there is little to no interaction of integrins and
other cell surface receptors with the gels.
7.2.2.2.2 Hydrogels
Crosslinking of water-soluble polymer chains produces
water-insoluble 3-D networks. The product is called
''hydrogel''. Several distinctive features, including tissue-
like viscoelasticity, diffusive transport, and interstitial
flow characteristics, make synthetic hydrogels excellent
physicochemical mimetics of natural ECMs and candi-
dates for soft tissue scaffolds. Indeed, hydrogels have
a potential to efficiently encapsulate cells and high water
contents to allow for nutrient and waste transport. Soft
tissues of our body are also like a hydrogel because of
their high water contents ranging between 70% and 90%.
The structural integrity of hydrogels depends on cross-
links formed between polymer chains via physical, ionic,
or covalent interactions. Synthetic hydrogels can be
processed under relatively mild conditions, have struc-
tural properties similar to ECM, and can be delivered in
the body in a minimally invasive manner, particularly by
injection. A variety of synthetic materials including PEG,
poly(vinyl alcohol) (PVA), and poly(acrylic acid) may be
used to form hydrogels. These polymers are not bio-
degradable but water soluble unless crosslinks are intro-
duced. Thus, once the introduced crosslinks are broken,
7.2.2.2.3 Others
Besides aliphatic polyesters and hydrogels, a number of
synthetic polymers have been used for scaffolds. The
chemical structures of these polymers are shown in
Fig. 7.2-11
. Only polyurethanes (PUs) and poly-
phosphazens will be described here, because these
two polymers have been studied by different research
groups.
1000
10,000
100
1000
Polyurethanes
A considerable majority of tissue engineering literature
has utilized a narrow array of polyester scaffolds although
their mechanical properties are limited with respect to
high strain, elastic capabilities. As a result, attention has
been paid on developing bioabsorbable elastomers with
high strain, elastic capabilities, and mechanical strength.
The impact of mechanical forces on tissue develop-
ment is increasingly appreciated in efforts to engineer
load-bearing and mechanically responsive tissues. The
polymers applicable for these tissues should address the
requirement to transmit mechanical cues to the tissues
over the course of tissue development, regardless
of
in vitro
mechanical training or dynamic
in vivo
movement. Segmented PU elastomers potentially meet
10
100
1
10
0.1
0
0.2
0.4
0.6
0.8
1
Mole fraction of
ε
-caprolactone in copolymers
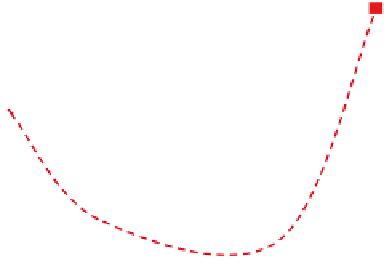
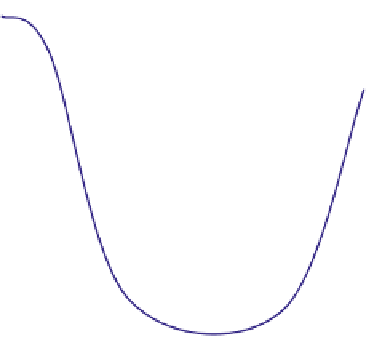
Fig. 7.2-10 Young's modulus and hydrolysis time until to 50%
reduction of tensile strength for LLA-
3
-CL copolymer plotted
against the mole fraction of
3
-CL comonomer.