Environmental Engineering Reference
In-Depth Information
Fig. 8
Mapping of PAA ISA
23 for DPD simulations
log
⊧
⊨
ʸ
+
pKa
1
,
pH
<
pKa
1
,
1
−
ʸ
log
ʸ
pH
=
+
pKa
2
,
pKa
1
<
pH
<
pKa
2
,
(46)
⊩
1
−
ʸ
log
1
1
+
pKa
3
,
pKa
2
<
pH
<
pKa
3
,
−
ʸ
where
ʸ
is the ratio between the number
N
−
of protonated-deprotonated monomeric
units and the total number
N
of monomeric units, and
pKa
i
is the acid-base equi-
librium constant. The variation of pH at constant ionic strength makes available the
control of the partial charge over the macromolecule. The DPD parameters
a
ij
are
calculated as described in Sect.
3
using the solubility parameters obtained by mole-
cular simulation. Ionic strength was fixed to 0
.
1
M
and the pH was varied according
to Eq. (
46
).
Performing electrostatic DPD simulations at different pH's, the mean radius of
gyration was calculated for 25 blocks of 10,000 steps. The size of the system was
L
x
=
L
y
=
L
z
=
8
.
5. Also,
ʳ
=
1
.
6 and
˃
=
3. PAA ISA 23 was represented by
48 DPD beads joined by springs with
k
=
2. The results as a function of pH and
ʸ
are shown in Fig.
9
.
According to these simulations, the PAA ISA 23 radius of gyration increases to
a maximum when the pH decreases. At high pH, and therefore high ionic strengths
(because of the counter-ions present in the system), the polymer is negatively charged
and adopts a rather compact structure. The conformation is displayed in Fig.
10
,
showing how the negative counter-ions (violet beads in the figure) are distributed
near the extreme of the polymer where the amide group is located and the internal
structure is extended at low pH (
ʸ
=
.
8333). At high pH (
ʸ
=−
.
75) the positive
counter-ions (orange DPD beads in the figure) are around the carboxyl extreme.
Experimental data reported by Griffiths et al. (
2004
) shows a very similar but more
complex equilibrium in the system: with decreasing pH, the PAA ISA 23 radius of
gyration increases to a maximum around pH = 3, after which value a decreasing
R
g
is
1
0





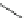










































Search WWH ::

Custom Search