Biology Reference
In-Depth Information
Table 2
DNA fragmentation at four autocatalytic rates
of caspases
DNA Fragmentation
Stop time rate0 rate1 rate2 rate3
10 0 0 0 1169
15 251 442 746 1862
20 417 581 885 2048
The autocatalytic rates are: rate0
=
0, rate1
=
mA
*
mB
/80000, rate2
=
mA
*
mB
/40000, and
rate3
=
mA*mB
/25000. They are assigned
to the transition
T
A
in Fig. 12. The stop
time represents the period after that Fas lig-
and stimulation is stopped. The initial Fas
ligand concentration is set to be
n
=
600.
Variables
mA
and
mB
represent the contents
of the continuous places going into
T
A
.
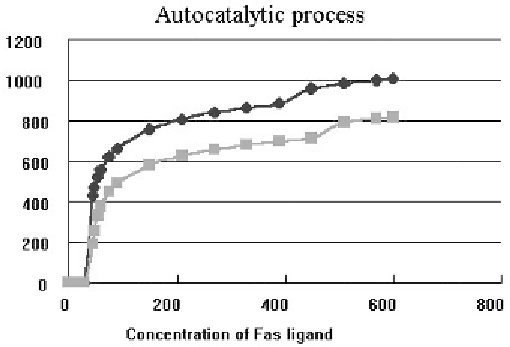
Fig. 11. Simulated relationship between the DNA fragmentation amount and the Fas ligand concentration: At higher concentra-
tion of Fas ligand, the direct pathway from caspase 8 to caspase 3 contributes to the fragmentation. To examine the effect of the
autocatalytic process of caspases, DNA fragmentation is simulated for both cases of the presence and absence in this process.
Since the presence of an autocatalytic process is proposed in caspase reactions [21], it has been included
in our model (Fig. 12), which increases the DNA fragmentation as shown in Fig. 11. However, if a high
rate of the autocatalytic process is assumed in the caspase reaction, the DNA fragmentation becomes
independent of the Fas ligand concentration, which does not coincide with the experimental results.
Therefore, we can guess that autocatalytic processes must be slow if they are present. To examine the
effect of autocatalytic processes of caspases on the apoptosis induced by Fas ligand, DNA fragmentation
is simulated when the stimulation by Fas ligand stopped after a short period. Table 2 shows a simulation
result that the apoptosis proceeds more with the increase of the autocatalytic rate of caspases even for a
short period stimulation.
Figure 13 shows simulated time courses of the HFPN in Fig. 10 with GON. Some intermediates during
apoptosis at three levels of Fas ligand concentrations are measured. These time courses might be useful
to plan new experiments such as addition of inhibitors to some step. However, it is necessary to estimate
the realistic rates of each reaction by the comparison with the experimental data. It is also necessary to