Chemistry Reference
In-Depth Information
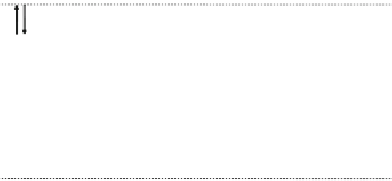
Fig. 12.4 Second-order
processes with respect to
t
MM
and
t
MXM
in the CDW and CP
phases
CDW
t
MM
U
M
X
X
t
MXM
CP
t
MM
2
β
|y|
X
X
t
MXM
Though the effects of the long-range interactions are discussed later, it is here noted
that
V
MM
increases the energy of the CDW phase and stabilizes the CP phase. The
second-order processes with respect to
t
MM
shown in Fig.
12.4
lower the energy by
2
t
MM
2
/
U
M
in the CDW phase and by
t
MM
2
=ð
2
bjyjÞ
in the CP phase. That is why the
CDW phase is relatively stable for large
and the CP phase for large
U
M
.Ina
similar manner, the effect of
t
MXM
can be discussed from the second-order pertur-
bation theory. It does not affect the relative stability between the CDW and CP
phases. That is why the phase boundary is insensitive to
t
MXM
. It is noted that, if the
long-range interactions are included, the second-order terms with respect to
t
MXM
for the CDW and CP phases become different. Even in such a case, the phase
boundary is more sensitive to
t
MM
.
Experimentally, the ground states of K
4
[Pt
2
(pop)
4
X]
n
H
2
O with X
b
¼
Cl, Br and
n ¼
2, 3 are shown to be in the CDW phase [
1
-
4
]. In general, when the halogen X
ion is either Cl or Br, so far all the ground states are known to be in the CDW phase.
For X
Cl and Br, the X p
z
level is so deep that the effective nearest-neighbor
transfer integral
t
MXM
mediated by charge transfers through the X p
z
orbital is
small. Meanwhile, the site-diagonal electron-lattice coupling
¼
is large owing to
the short distance between the neighboring M and X ions,
d
MX
. Thus, the CDW
phase for X
b
¼
Cl and Br is understood from the dominance of the electron-lattice
coupling
over electron-electron interactions other than the on-site repulsion
U
M
,
which is strong enough to suppress a phase with bipolarons discussed later.
In R
4
[Pt
2
(pop)
4
X]
n
H
2
O with X
b
I, depending on the cation R and on the
number of water molecules
n
, three electronic phases are suggested to appear: the
AV phase, the CDW phase, and the CP phase in the order of increasing of
d
MXM
[
7
].
Though we have not obtained a phase diagram with the CDW phase between the
other two, we believe that, for small
d
MXM
, the effect of
t
MXM
is larger than those
of various interactions so that the larger
t
MXM
stabilizes the AV phase. Meanwhile,
for intermediate to large
d
MXM
, the competition between the electron-lattice
and electron-electron interactions discussed here and/or the competition between
the short- and long-range electron-electron interactions discussed later determine
the relative stability between the CDW and CP phases. As
d
MXM
increases, the site-
diagonal electron-lattice coupling
¼
b
becomes weak, while the on-site repulsion


























Search WWH ::

Custom Search