Chemistry Reference
In-Depth Information
Pt
Pt
Pt
c
b
Pt Cl
CN
O
H
a
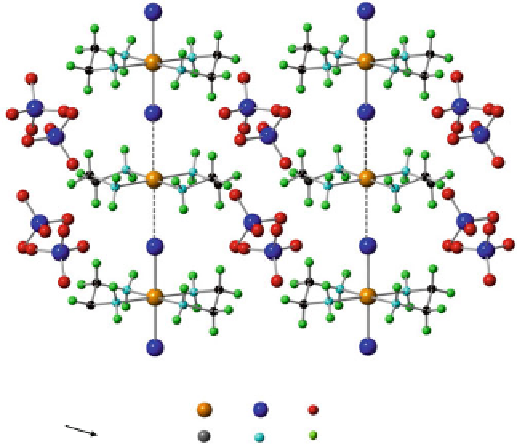
Fig. 2.1 Structure of [Pt(en)
2
][Pt(en)
2
Cl
2
](ClO
4
)
4
their electronic states are not so controllable. Therefore, it is difficult to perform
systematic studies of excitons, solitons, and polarons in those systems.
Subsequently, halogen(X)-bridged transition metal(M) compounds (or equivalently
MX-chain compounds) have provided a unique opportunity to study the nature of
excitons, solitons, and polarons [
5
]. A great advantage of this category of materials is
that their electronic structures can be widely controlled and a variety of materials can be
obtained as single crystals. As a result, a number of studies concerning excitons,
solitons, and polarons have been reported so far in MX-chain compounds [
6
-
52
].
MX-chain compounds are represented as [MA
2
][MA
2
X
2
]Y
4
(or simply {MA
2
X}
Y
2
), where A and Y are the ligand and the counter anion, respectively. The crystal
structure of [Pt(en)
2
][Pt(en)
2
Cl
2
](ClO
4
)
4
(en = ethylenediamine) is shown in Fig.
2.1
as a typical example of MX-chain compounds. As shown in the figure, the [MA
2
]
moieties are bridged by the halogen ions (X) and the hydrogen (H)-bonds between
the amino groups of the ligands (A) and the counter anions (Y) support the MX
chains. In the compounds with M
¼
Pt and Pd, M(II) and M(IV) mixed-valence
state, or equivalently, the commensurate charge-density-wave (CDW) state is
stabilized due to the strong e-l interaction, while in the compounds with M
Ni,
Ni(III) mono-valence state, or equivalently, the Mott-Hubbard (MH) state appears
synthesized.
In the Pt and Pd compounds, amplitudes of CDW can be controlled by the
replacements of metals (M
¼
¼
Pt and Pd), bridging halogens (X
¼
Cl, Br, and I),
ligand molecules (A
ethylenediamine (en), ethylamine, diaminocyclohexane
(chxn), etc.) and counter anions (Y
¼
ClO
4
,BF
4
, Cl, Br, I, etc.) surrounding the
MX chains [
28
,
32
]. In addition, the nondegenerate CDW states can be obtained in
¼


Search WWH ::

Custom Search