Biomedical Engineering Reference
In-Depth Information
environmental movement pointed out that there were environmental costs associated with
the many material benefits that were now being enjoyed. In the late twentieth century, envi-
ronmental problems became global in scale. The 1973, 1979, and 2008 energy crises demon-
strated the extent to which the global community had become dependent on nonrenewable
energy resources. While the use of fossils inadvertently changed the sustainable state, or
more accurately is making the transition to a different sustainable state, fossil use itself is
not sustainable. In the twenty first century, there is increasing global awareness of the threat
posed by the human-induced enhanced greenhouse effect, produced largely by forest
clearing for urban development and agriculture, and the use of fossil fuels.
Sustainability is studied and managed over many scales (levels or frames of reference) of
time and space and in many contexts of environmental, social, and economic organization.
The focus ranges from the (seemingly quantitative but subjective and able to manipulate)
total carrying capacity (sustainability) of planet Earth to the (qualitative) sustainability of
economic sectors, ecosystems, countries, municipalities, neighborhoods, home gardens, indi-
vidual lives, individual goods and services, occupations, lifestyles, behavior patterns, and so
on. In short, it can entail the full compass of biological and human activity or any part of it.
15.3. WATER
We have taken water as granted: it is vastly available on earth. Water covers 70.9% of the
Earth's surface. Water had been the cause of destruction and prosperity for ancient civiliza-
tions. Legends have it how our ancestors tamed the waterways and made land fertile to
agriculture.
Water is dependent on by all known forms of life. It is the best solvent and energy carrier
for humanity. The importance of water is due to its physical and chemical properties.
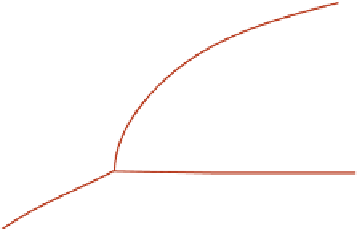
Fig. 15.4
shows the property of water. What makes water special is that under normal atmospheric
(a)
(b)
Critical point
374° C
Steam
Boiling
100° C
Liquid Water
Triple point
Melting
0° C
Ice
1
218
Energy
Pressure, atm
FIGURE 15.4
Phase change of water with temperature and pressure. (a) Phase diagram of water. (b) Internal
energy change of water with temperature at 1 atmosphere.





















Search WWH ::

Custom Search