Biomedical Engineering Reference
In-Depth Information
Cu
2
C
2
H
5
OH
þ
O
2
!
2
CH
3
CHO
þ
2
H
2
O
Ag
2
CH
3
OH
þ
O
2
!
2
HCHO
þ
2
H
2
O
Pt
4
NH
3
þ
5
O
2
!
4
NO
þ
6
H
2
O
Ni
2
C
2
H
6
þ
7
O
2
!
4
CO
2
þ
6
H
2
O
5.
Hydration and dehydration (or condensation). This type of reactions are catalyzed by
substances that have strong affinity to water. Al
2
O
3
, SiO
2
-Al
2
O
3
gel, MgO, clay,
phosphoric acid, and salts are good catalysts. For example,
CH
3
CH
2
OH
which is an interesting reaction. It used to be a cheap way to produce ethanol from petro-
leum-based ethylene. The reverse of the reaction is more important today as ethanol can
be produced from renewable resources, and ethylene is a valuable monomer for higher
alkenes (jet-fuel) and polymer production.
6.
Halogenation and dehalogenation. These reactions are commonly catalyzed with an
active ingredient of CuCl
2
, AgCl, or Pd. Halogenation can occur without catalyst.
However, catalyst is used to improve selectivity.
CH
2
]
CH
2
þ
H
2
O
/
Solid catalysts usually have fine pores such that reactants can be “fixed” or brought tightly
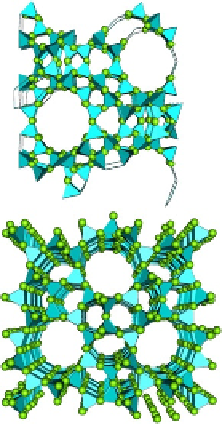
together by active centers on the surface for reaction to occur.
Fig. 9.1
shows a schematic of
typical zeolite catalysts.
12-Member
Ring
Faujasite-Type
Zeolite
7.4Å
10-Member
Ring
6.6Å
ZSM-5 Zeolite
FIGURE 9.1
Typical zeolite (alumina-silica or Al
2
O
3
-SiO
2
) catalyst: 3-D pore formed by 12 oxygen ring and 1-D
pore formed by 10 oxygen ring. More zeolite structures can be found at
http://www.iza-structure.org/databases/
.

























Search WWH ::

Custom Search