Biomedical Engineering Reference
In-Depth Information
Fig. 4.2
Quanti fi cation of
the proliferation status of
HDMEC grown on PS and
Ti6Al4V 24 h after treatment
with H
2
O
2
as assessed by
measuring Ki67 expression
related to the cell number
(means ± SDs; untreated
control on PS set as 100 %,
signi fi cant
difference:*
p
< 0.05,
**
p
< 0.01)
140
PS
Ti6AI4V
*
**
120
100
80
60
40
20
0
0
0.25
0.5
H
2
O
2
(mM)
patients with failed and stable Ti6Al4V alloys, while the concentration of V remained
low [
36
]. An increased metal ion concentration can be detected even in individuals with
no complications after implantation of Ti-based prostheses, suggesting that metal cor-
rosion occurs even in the absence of metal wear debris formation [
37
] .
The cathodic part of the corrosion process, in contrast, results in the reduction of
oxygen at physiological pH with the formation of ROS and H
2
O
2
as intermediate
products [
59
]. Thickening of the TiO
2
layer in biological solutions substantiates the
fact that corrosion processes permanently occur at titanium implant surfaces [
49,
66
]. Besides the possible formation of ROS by the titanium (-alloy) itself as the
result of cathodic corrosion, titanium may be subjected to the ROS produced by
inflammatory cells coming into contact with titanium (-alloy) surfaces directly after
implantation. One of the mediators is H
2
O
2
released by monocytes, macrophages
and granulocytes. The TiO
2
layer may interact with H
2
O
2
leading to formation of
hydroxyl radicals [
44
]. Altogether, these facts led to the hypothesis that endothelial
cells that take part in wound healing early after implantation might be permanently
subjected to ROS formed at the titanium implant surface, which exceeds physiolog-
ical protection mechanisms and can thus be referred to as oxidative stress. To
confirm this hypothesis the reactions of endothelial cells to the Ti6Al4V alloy in the
presence of the oxidative stress inducer H
2
O
2
in comparison to the reactions elicited
in endothelial cells grown on cell culture PS were studied.
The quantification of human dermal microvascular endothelial cells (HDMEC)
growing on Ti6Al4V and PS revealed the dose-dependent reduction of cell number
to the same extent on PS and Ti6Al4V alloy 24 h after H
2
O
2
treatment compared to
the untreated control, with no significant differences between the materials [
87
] . In
contrast, expression of Ki67, a marker for cell proliferation, was lower in HDMEC
grown on Ti6Al4V alloy compared to the cells grown on PS (Fig.
4.2
). This pointed
to the possibility of higher H
2
O
2
cytotoxicity on Ti6Al4V alloy. It has been shown in










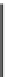





















Search WWH ::

Custom Search