Environmental Engineering Reference
In-Depth Information
the Langmuir-Hinshelwood (L-H) model as described by Zhou and
Ray [40].
dC
dt
V
V
kKC
KC
(4.1)
r
b
L
r
s
rxn
1
R
s
where V
L
is the total volume of liquid treated (m
3
), V
R
is the volume
of the reactor (m
3
), (dC
b
/dt) is the observed rate (mol.m
-3
.s
-1
), k
r
is the
reaction rate constant (mol.m
-3
.s
-1
), K is the adsorption-desorption
equilibrium constant (m
3
.mol
-1
), C
s
is the concentration of the reactant
on the catalyst surface (mol.m
-3
) in equilibrium with the actual surface
concentration [40].
A Langmuir-type kinetic equation is more appropriate for photocata-
lyst slurry system; in case of immobilized system it is difficult to predict
the kinetics without knowing the pollutant concentration on photocatalyst
surface. Zhou and Ray [40] considered both external and internal mass-
transfer resistance for photocatalytic degradation of Eosin B on immobi-
lized TiO
2
surface to find out the true kinetic parameters. They found out
the intrinsic kinetic parameters from the following equation:
11
1
1
(4.2)
k
k
k
k
k
k
o
r
mint
,
mext
,
where k
r
is the first-order reaction rate constant (s
-1
), k
m,ext
is external
mass-transfer coefficient, and k is the specific surface area (m
-1
).
4.6.3
Effect of Solution pH
The pH of the solution significantly affects a photocatalytic process as it
occurs on the surface of the photocatalyst. Various surface properties of
photocatalyst such as: i) surface charge, and ii) band edge position, are
influenced by pH. Degussa P25 TiO
2
shows point of zero charge (pHzpc) at
pH 6.8, and thus in alkaline medium, the TiO
2
surface becomes negatively
charged. Therefore in alkaline pH, cationic dye (e.g., methylene blue) is
well adsorbed [40,41,42].
IV
IV
Ti
OH
HO
Ti
O
H O
2
IV
IV
Ti
OH
H
Ti
OH
2
Zhou and Ray [40] performed a kinetic study for photocatalytic degra-
dation of an anionic dye (Eosin B) with TiO
2
. The adsorption of Eosin B



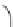

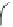

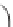





















Search WWH ::

Custom Search