Biomedical Engineering Reference
In-Depth Information
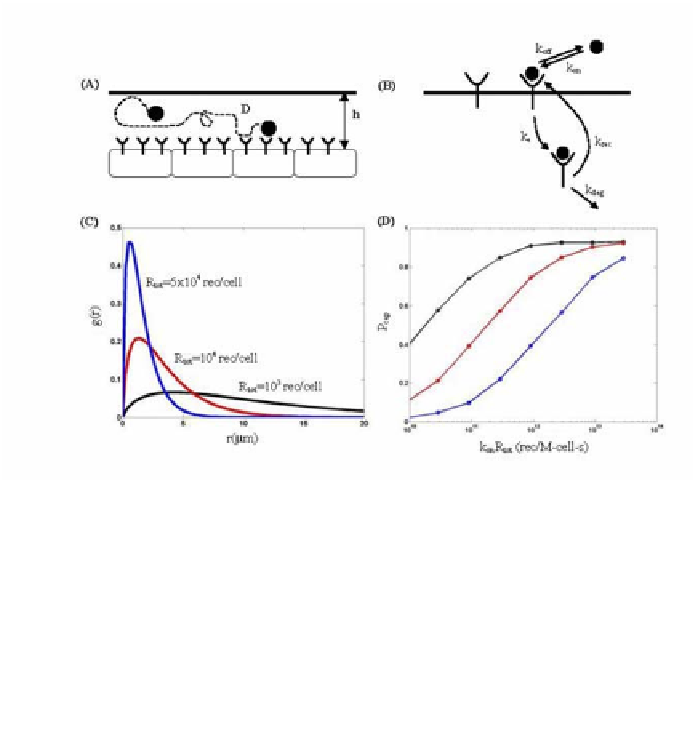
Figure 6
. (
A
) A simplified model of ligand transport, binding, and trafficking. Ligand diffuses
in the gap between the reflective and receptor-covered surfaces. Receptor density is uniform
across the surface of the epithelial layer. (
B
) Ligand-receptor interactions (see Table 1 for
definition of parameters). (
C
) Probability density function for the lateral distances traveled by
secreted ligands in the time between the binding events.
g
(
r
)
dr
is equal to the probability that a
ligand will be bound between
r
and
r
+
dr
(see Eq. [2]). All computations are performed on a
hexagonal cell with an area of 25
m
2
. (
D
) Fraction of the ligands that are recaptured by the
ligand-releasing cell plotted as a function of the cell surface receptor number (
R
tot
), ligand-
receptor affinity (
k
on
), and extracellular ligand diffusivity (
D
). The curves, from top to bottom,
correspond to
D
= 10
-9
cm
2
/s,
D
= 10
-8
cm
2
/s, and
D
= 10
-7
cm
2
/s.
effective diffusion coefficient D. The diffusion coefficient can vary between the
low values of growth factor diffusion in extracellular matrices (10
-10
cm
2
/s) (37)
and the typical values for protein diffusion in an aqueous solution (10
-6
cm
2
/s)
(38). We assume that the number of receptors per cell,
R
tot
, is constant. As illus-
trated in Figure 6B, ligand-receptor interactions are characterized by kinetic rate
constants
k
on
and
k
off
; the endocytosis of receptor-bound ligands is modeled as a
first-order process with rate constant
k
e
; we assume that internalized ligand is not
recycled. The last assumption is based on the observation that, for the mammal-
ian TGF
, recycling is negligible (39). In the absence of measurements in the
Drosophila
EGFR system, the rate constants are approximated by their counter-
parts measured in mammalian systems (39,40). In the following, we show how
this model can be used to quantify the distance traveled by a secreted ligand.
Analysis of the distance traveled by a ligand between the subsequent bind-
ing events requires solving the problem of ligand transport in the gap above the