Environmental Engineering Reference
In-Depth Information
Integration of the above equation with the boundary conditions
t
= 0,
q
= 0, and at
t
=
t
,
q
=
q
, results in:
1
1
kt
qqq
=+
(11.9)
2
−
e
e
h is equation can be stated in the linear form as:
t
t
qq
kq
1
=+
2
2
(11.10)
e
e
where
k
2
is the equilibrium rate constant of pseudo-second-order adsorp-
tion (g/mg/min). h e pseudo-second-order rate constants can be deter-
mined experimentally by plotting
t
/
q
against
t
. As such, in comparison
to the pseudo-i rst-order kinetic model, this model is considered more
appropriate to represent the kinetic data in adsorption systems [104].
Pseudo-i rst-order and pseudo-second-order rate expressions have
been and still are in wide use for studying the adsorption of heavy metals
from aqueous solutions. In the chemisorption process, the pseudo-second-
order is superior to the pseudo-i rst-order model, as it takes into account
the interaction of adsorbent-adsorbate through their valency forces [104].
11.7.1.3
Weber and Morris Sorption Kinetic Model
h e Weber and Morris sorption kinetic model [109] was initially employed
by Pasavant
et al.
[110] to describe their biosorption experimental data.
h is model has the following form:
qK t
=
(11.11)
WM
In their investigation, the sorption process by
C. lentillifera
biomass for
Cu(II), Cd(II), Pb(II), and Zn(II) was regulated by two main mechanisms:
intraparticle dif usion and external mass transfer. Intraparticle dif usion
can be estimated with:
2
dK
pWM
D
=
(11.12)
8640
q
e


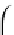






Search WWH ::

Custom Search