Biology Reference
In-Depth Information
X-ray structural analyses of
Bj
FixLH reveal that O
2
binding to the haem
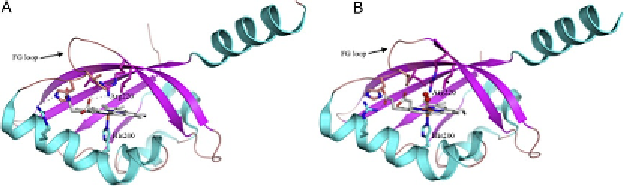
induces a shift of the FG loop (Thr209 to Arg220) close to the haem pocket
(
Fig. 7.4
). This shift results in the formation of a hydrogen bond between the
haem-bound O
2
and Arg220. O
2
-binding affinity decreases by about
10-fold when Arg220 is replaced by Ala, indicating that the hydrogen-
bonding interaction of Arg220 with the haem-bound O
2
plays an important
role for the regulation of O
2
-binding affinity (
Table 7.1
).
Though Arg220 forms a salt bridge with the haem propionate 7 in deoxy
Bj
FixLH, this salt bridge is dissociated upon O
2
binding and the guanidium
side chain of Arg220 rotates into the distal haem pocket to form the hydrogen
bond with the haem-bound O
2
. The C
a
d
C
b
bond of Arg220 is rotated with
ca. 170
(
Gong et al., 2000
). There is a hydrogen-bond network among
Arg220, O
2
, a water molecule, and the carbonyl oxygen of Ile218, which will
be stabilised by the proper orientation of Arg220 in oxy
Bj
FixLH.
While CN
binding causes a similar FG loop shift, such an FG loop shift
does not take place upon CO or NO binding (
Gong et al., 2000; Hao et al.,
2002
). In CN
-bound
Bj
FixLH, Arg220 moves into the haem pocket to
form an electrostatic interaction with the haem-bound CN
, while the salt
bridge between Arg220 and the haem propionate retains in CO- and
NO-bound
Bj
FixLH. The structure of Im-bound
Bj
FixLH reveals changes
in the FG loop that are similar to those observed in oxy and CN
-bound
Bj
FixLH, though Arg220 adopts a position outside of the haem pocket
(
Gong et al., 2000
). Both CN
and Im can regulate the FixL activity as does
O
2
. These results indicate that the interaction between the side chain of
Arg220 and the haem-bound ligand is not a mandatory requirement for
the regulation of FixL activity.
Based on these results, “the FG loop mechanism” is proposed for the reg-
ulation of FixL activity. In this mechanism, it is proposed that O
2
binding to
Figure 7.4 The structures of (A) met BjFixLH (PDB 1DRM) and (B) oxy BjFixLH (PDB
1DP6).