Environmental Engineering Reference
In-Depth Information
[
O
3
]
∗
=
P
O
3
/K
aw
, where
K
aw
=
0.082 is the Henry's constant expressed in
atm m
3
/mol. For a typical
P
O3
=
O
3
]
∗
=
0.092 mol/m
3
. Hence
ss
0.0075 atm,
[
[
O
3
]
=
10
−
4
mol/m
3
. A mass balance for ozone over the entire reactor yields
9
×
Q
G
ou
g
i
g
ss
[
O
3
]
−[
O
3
]
=
(Q
L
+
k
tot
V
L
)
[
O
3
]
=
4 mol/min.
If
Q
G
=
1000 m
3
/h
(
=
16 m
3
/min),
[
O
3
]
i
g
out
g
=
0.24 mol/m
3
. Since
[
O
3
]
i
g
−[
O
3
]
=
P
O
3
/RT
=
0.31 mol/m
3
, we obtain
[
O
3
]
out
g
=
0.07 mol/m
3
. Hence the ozone transfer
efficiency in the reactor is 77%.
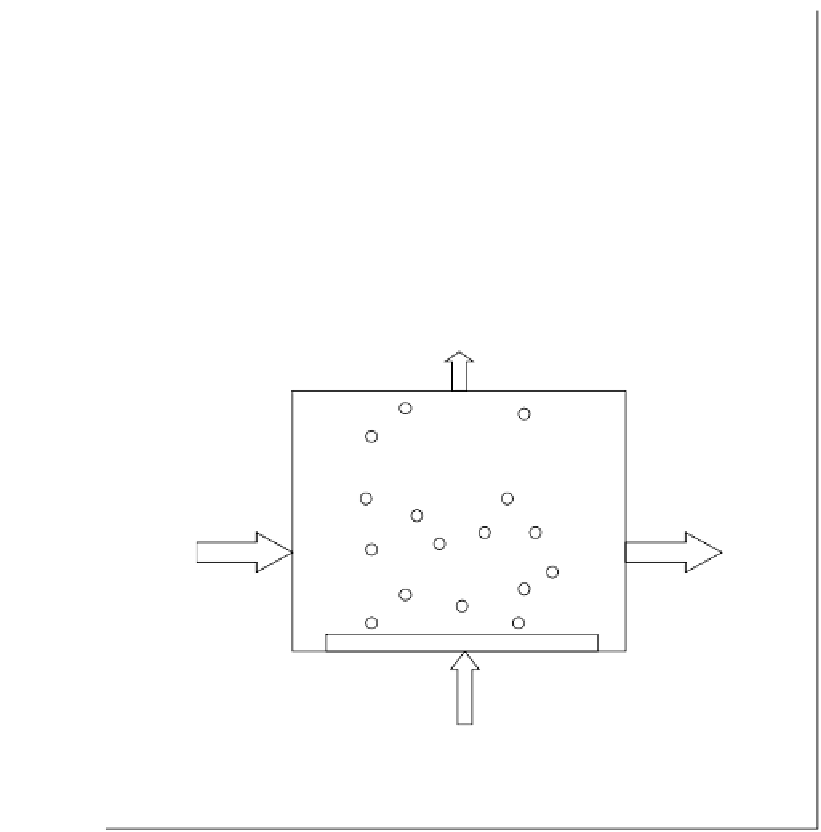
Q
G
[O
3
]
out
water
[O
3
]
M → M
ox
Q
L
Q
L
[O
3
]
[O
3
]
in
= 0
[M]
in
[M]
out
Ozone
Q
G
[O
3
]
in
FIGURE 6.23
Schematic of an ozone reactor for wastewater oxidation.
6.2.2.3
Photochemical Reactions and Wastewater Treatment
Photochemical reactions are useful in treating wastewater streams. An application in
this area is the use of semiconductors (e.g., TiO
2
) in enhancing the UV-promoted oxi-
dation of organic compounds. The reaction pathway provided by TiO
2
is complicated
(Legrini, Oliveros, and Braun, 1993). TiO
2
is a semiconductor. It has a structure that
is composed of a valence band (filled electronic level) and a conduction band (vacant
electronic level) that are separated by a band gap (see Figure 6.24). As an electron
jumps from the valence band to the conduction band, a hole (positive charge) is left
behind in the valence band. This
e
−
jump can be brought about through excitation by
light (UV or visible). Organic molecules that are thereby oxidized can scavenge the
hole left behind in the valence band. The photoexcited TiO
2
with the electron-hole








Search WWH ::

Custom Search