Environmental Engineering Reference
In-Depth Information
1-Diffusion of reactant
2-Surface adsorption
3-Surface reaction
4-Product desorption
5-Diffusion of product
A
P
1
5
2
4
3
Z
P
Solid surface
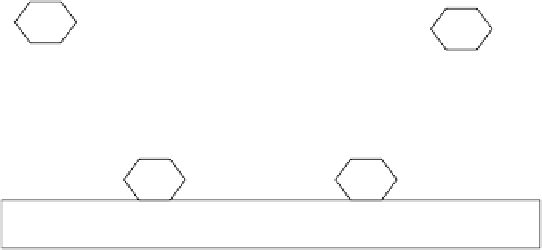
FIGURE 5.12
Schematic of the steps in a heterogeneous reaction on the surface of a solid.
A heterogeneous surface reaction mechanism involves postulating that a molecule
(A) becomes adsorbed on a surface (X), which further becomes an activated complex
(Z) that then breaks down to give the product (P). This is the basis of the
Langmuir-
Hinshelwood
mechanism for heterogeneous surface reactions.
A
+
X
Z
−→
X
+
P.
(5.133)
If two species A and B are involved we have the following scheme:
A
+
X
Z
1
,
B
+
X
Z
2
,
(5.134)
Z
1
+
Z
2
−→
X
+
P.
The above scheme requires that two species be adsorbed on adjacent surface sites. In
some cases, only one (say B) gets adsorbed which then reacts with a gaseous species
(say A) to give the products. This is the
Langmuir-Rideal
mechanism.
For the Langmuir-Hinshelwood mechanism, the rate of the reaction depends on
the surface concentration of A. The Langmuir isotherm for adsorption from the gas
phase gives the surface coverage of A,
K
Lang,A
P
A
θ
A
=
,
(5.135)
1
+
K
Lang,A
P
A
The rate of conversion of the adsorbed complex to products is
K
Lang,A
P
A
r
=
k
θ
A
=
k
.
(5.136)
1
+
K
Lang,A
P
A
At high pressures,
K
Lang,A
P
A
1,
r
→
k
and is independent of the concentration
of A. At low pressures,
K
Lang,A
P
A
1 and
r
→
kK
Lang,A
P
A
and the rate is first
order in A.













Search WWH ::

Custom Search