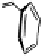Biology Reference
In-Depth Information
Prior to and beyond such considerations of strict analytical work
concerns, the most important issue that should be kept in mind at the
scientific level is the unveiling of a given type of chemistry at play in a
given system. So, not only vescalagin (
1
), but any
C
-glycosidic
ellagitannins displaying a free hydroxyl group at C-1, such as roburin A
(
5
), stachyurin (
3
), 5-desgalloylstachyurin (
26
) and also vescalin (
33
)
(Mayer
et al.
, 1967, 1971b) can thus act like “nucleophile sponges” if
present, introduced or generated in wine. Vescalin (
33
) naturally results
from the hydrolysis of the 4,6-HHBP group of vescalagin (
1
) (Fig. 9.12).
It is present in the heartwood of oak species used for cooperage (Viriot
et
al.
, 1994), albeit in rather small amounts estimated to range between 0.11
and 1.48 mg/g of dry wood (Masson
et al.
, 1995, 1996, Mosedale
et al.
,
2001), but can thus end up in the wine solution (Moutounet
et al.
, 1989,
Puech
et al.
, 1996), in which hydrolysis of
1
into
33
can also occur.
Additional examples of the expression of this reactivity between
ellagitannin C-1 benzylic alcohols, again exemplified by vescalagin (
1
),
and other wine nucleophiles are highlighted in the following sections.
OH
OH
R
1
1
4
6
O
O
O
O
OH
HO
33
: R
1
=
β
-OH, vescalin
34
: R
1
=
α
-OH, castalin
O
O
HO
OH
HO
OH
HO
OH
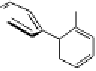
Fig. 9.12 Stuctures of vescalin (
33
) and castalin (
34
) derived from the hydrolytic
removal of the 4,6-HHBP group in vescalagin (
1
) and castalagin (
2
), respectively.
HO
9.3.2 An example of a condensation reaction with a non-phenolic
wine nucleophile
The condensation reaction of vescalagin (
1
) with the cysteine-containing
tripeptide glutathione (
35
) was also investigated. This choice of
nucleophile originates from observations made by enologists on the
decrease of the level of
35
in wines, especially white wines, aged in new
oak barrels (Lavigne-Cruege
et al.
, 2003). These observations were
particularly relevant to the quality control of white wines, for glutathione