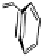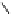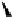Biology Reference
In-Depth Information
Calculations of the lowest-energy unoccupied molecular orbital
(LUMO) of
27
showed a large and symmetrical sp
2
-hybridized atomic p-
orbital at C-1. Access of a nucleophile to C-1
via
the α-face of this
vacant cationic p-orbital is probably hindered by the steric encumbrance
of the
endo
-face of the molecule. Most significant was the observation of
a larger electron-deficiency on the “reacting”
exo
β-face of the orbital by
mapping the LUMO onto the electron density surface of
27
(Fig. 9.9).
HO
exo
β
-face
endo
α
-face
HO
OH
HO
O
O
HO
O
O
HO
27
O
1
O
O
O
OH
HO
O
O
HO
OH
II
HO
OH
OH
HO
HO
HO
HO
OH
HO
O
O
HO
O
O
HO
27'
O
O
O
O
OH
HO
H
H
O
HO
OH
II
HO
OH
HO
OH
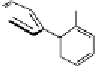
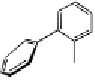
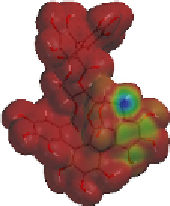
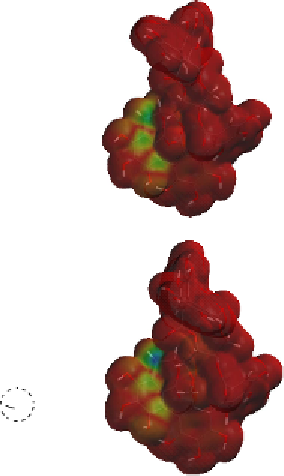
Fig. 9.9 Facial views of mapping of Spartan-generated Hartree-Fock models of the
LUMO of vescalagin-derived benzylic cations
27
and
27'
onto their corresponding 0.002
electron au
-3
electron density isosurfaces. Replacement of the carbonyl group of the
NHTP galloyl-derived II-ring of
27
by a methylene unit (see dotted circle on structure
27'
) results in a recovery of the elecron-deficiency of the
endo
α-face of the vacant
cationic p-orbital at C-1. According to the color code used, the bluer the color, the more
electron-deficient the orbital is.
HO