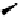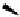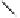Biology Reference
In-Depth Information
O
O
OH
CH
3
H
2
N
H
HN
NH
2
HO
HO
H
HN
NH
N
N
8
N
N
H
OH
H
3
C
CH
3
O
OH
O
O
OH
O
ouabain (
23)
.
4HCl
H
3
C
N
N
H
3
C
OH
3
HN
NH
2
H
2
N
NH
OH
CNI-1493 (
26
)
NH
NH
Fig. 6.7 Some LPS antagonists that act through NF-κB or p38 MAPK inhibition.
Ouabain (
23
), a cardiotonic
steroidal derivative, was reported to
suppress TNFα levels in LPS stimulated mice by targeting the Na,K-
ATPase enzyme, which is responsible for controlling intracellular
calcium and other cation levels, through an unidentified mechanism
involving NF-κB (Matsumori
et al.
, 1997, Bereta
et al.
, 1995).
Curiously, in the absence of LPS, ouabain (
23
) induced production of
TNFα. Selective inhibitors of NF-κB itself have not been identified, as
other kinases and proteases are also affected.
The stress-activated protein kinase p38/SAPK-2, a mitogen-activated
protein kinase (MAPK) that is responsible for releasing a translation
repressor protein complex from TNFα mRNA to allow protein synthesis,
also has been explored as a target for inhibition. The cytokine-
suppressing anti-inflammatory drugs (CSAIDS), which include
imidazoles SKF86002 (
24
) and SB210313 (
25
), target p38 MAP kinase.
Tetravalent guanylhydrazone CNI-1493 (
26
) also suppresses the
production of TNFα, perhaps through inhibition of the p38 MAP kinase
pathway (see Fig. 6.7).
TNFα-converting-enzyme (TACE), the enzyme responsible for
converting pro-TNFα into soluble TNFα, has been a hot target for anti-
TNFα therapy. TACE is a member of the same protease family
(metzincin), as the matrix metalloproteinases (MMPs). Thus, several
known MMP inhibitors have been modified to make selective TACE