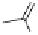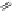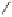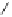Biology Reference
In-Depth Information
benzylidene acetal group of the required (
S
)-diastereoisomer
82
was then
cleaved under acid-catalyzed conditions and the resulting diol
83
was
acylated with enantiopure (
S
)-hexamethoxydiphenoyl dichloride (
S
)-
80
to furnish trideca-
O
-methyl-α-pedunculagin (
84
, Fig. 5.16).
O
Ph
O
O
O
BnO
O
O
BnO
O
OMe
OMe
BnO
R
MeO
81
BnO
O
Ph
OBn
O
BnO
HO
O
MeO
COCl
COCl
DMAP
+
+
MeO
81
22%,
82
23%
HO
O
Ph
OMe
O
51
O
MeO
rac
-
80
O
MeO
2
3
O
O
OMe
MeO
OMe
O
MeO
S
82
MeO
MeO
OMe
OMe
MeO
MeO
HCl, MeOH,
83%
MeO
S
O
O
HO
6
O
O
HO
MeO
4
O
O
O
(S)-
80
, Et
3
N
MeO
2
3
O
O
O
MeO
MeO
MeO
OMe
2
O
1
37%
3
O
O
MeO
OMe
O
MeO
S
83
MeO
MeO
S
OMe
MeO
MeO
OMe
MeO
84
: trideca-
O
-methyl-
α
-pedunculagin
Fig. 5.16
Synthesis of trideca-
O
-methyl-α-pedunculagin (
84
).
5.2.3.2
Total synthesis of pedunculagin via the biaryl coupling strategy
Two different strategies have been developed for the total synthesis of
pedunculagin (
87
). In the first strategy, the biaryl part of pedunculagin
was constructed by a diastereoselective oxidative coupling of phenolic
galloyl moieties attached to the
D
-glucopyranose core (Fig. 5.17, and see
Fig. 5.1, method
B
) (Feldman and Smith, 1996). The second strategy
relied on the double esterification approach (see Fig. 5.1, method
A
),
through which an appropriately protected diphenic acid derivative is