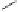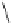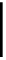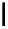Biology Reference
In-Depth Information
5.2.2.3
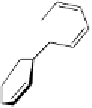
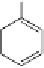
Construction of a 4,6-bridged ellagitannin scaffold
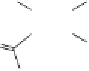
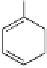
A copper-catalysed reductive Ullmann coupling of two methyl ether-
protected iodinated galloyl moieties attached to the appropriate positions
of a
D
-glucopyranosyl unit (see
39
in Fig. 5.8) produced the coupled (
S
)-
configured product
40
, together with the non-coupled and reduced
product
41
, in a 1:1 ratio. As in the case of the Feldman coupling, which
also corresponds to an application of method
B
(see Fig. 5.1) to construct
ellagitannin HHDP units, this reductive coupling reaction was also (
S
)-
atropodiastereoselective, as a consequence of conformational preferences
in
D
-glucopyranose (Dai and Martin, 1998).
OMe
OMe
I
OMe
MeO
I
O
OMe
O
O
MeO
6
4
O
O
BnO
BnO
OMe
39
MeO
OMe
OMe
Cu
40
/
41
= 1:1 ratio
OMe
MeO
OMe
MeO
MeO
O
O
OMe
+
O
MeO
O
O
O
MeO
6
O
O
4
MeO
O
BnO
O
BnO
BnO
OMe
BnO
40
OMe
41
Fig. 5.8 Construction of the 4,6-HHDP-containing ellagitannin framework.
5.2.2.4
Synthesis of O-perbenzyllagerstannin C
The isolation of lagerstannin C (
47
) from the fruit and leaves of
Lagerstroemia speciosa
(L.) Pers. (L.
flosreginae
RETZ.) (
Lythraceae
)
was first reported in 1992 along with that of some other lagerstannins
featuring a
D
-gluconic acid unit as a special structural characteristic
(Tanaka
et al.
, 1992b). It is well known that the tannin-rich extracts of