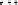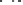Biology Reference
In-Depth Information
O
O
O
O
H
- H
2
O
OH
HO
OH
O
O
OH
OH
HO
HO
OH
OH
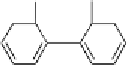
hexahydroxydiphenoyl group
(HHDP)
+ H
2
O
O
O
H
O
O
HO
H
HO
OH
O
OH
O
HO
O
OH
HO
O
OH
OH
OH
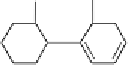
dehydrohexahydroxydiphenoyl group
(DHHDP)
Fig. 4.9 Structures of HHDP and DHHDP groups.
Some specific dehydroellagitannins, such as geraniin (
16
, 1-
O
-
galloyl-2,4-(
R
)-DHHDP-3,6-(
R
)-HHDP-β-
D
-glucose) (Okuda
et al.
,
1982), are accumulated in high concentration in some plants, such as
those of the families
Euphorbiaceae
,
Geraniaceae
(Okuda
et al.
, 1980),
and
Elaeocarpaceae
(Tanaka
et al.
, 1986). In many cases, the tannins are
accompanied by structurally-related ellagitannins having their acyl
groups biogenetically derived from the DHHDP group. Typical
examples are the chebuloyl (Schmidt and Mayer, 1951, Yoshida
et al.
,
1980, Yoshida
et al.
, 1982, Lin
et al.
, 1990a, Nonaka
et al.
, 1992),
brevifolin carboxyl, dehydrochebuloyl (Saijo
et al.
, 1989a, Lin
et al.
,
1990b), tetrahydroxydibenzofuran dicarboxyl (Saijo
et al.
, 1989b),
biscyclohexenetrionyl (Nonaka
et al.
, 1990), elaeocarpusinoyl (Tanaka
et
al.
, 1986), putranjivainoyl (Lin
et al.
, 1990a) and jolkinoyl (Lee
et al.
,
2004) groups (Fig. 4.10). The unique and attractive chemical structures
of these acyl groups reveal the complexity of the oxidative metabolism
of the HHDP groups. Of particular note is the fact that the oxidation
states of the tetrahydroxydibenzofuran dicarboxyl and chebuloyl groups
are the same as those of the HHDP and DHHDP groups, respectively.