Biology Reference
In-Depth Information
been isolated in the meantime, providing evidence that this reaction type
is of general importance in plants (see references in Gross, 1989).
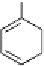
COOH
OH
HO
OH
+ UDP-Glc
- UDP
O
HO
O
HO
OH
HO
OH
OH
OH
O
1
2
Fig. 3.4 Biosynthesis of β-glucogallin (1-
O
-galloyl-β-
D
-glucopyranose,
2
) from free
gallic acid (
1
) and “activated” glucose, uridine-5'-diphosphate glucose (UDP-Glc). UDP
= uridine-5'-diphosphate.
3.5 'Simple' Galloylglucose Esters: the Route to Pentagalloylglucose
In the above discussed investigations on the biosynthesis of β-
glucogallin (
2
) with cell-free extracts from oak leaves, it was observed
that these enzyme preparations also catalyzed the transformation of
in
situ
generated β-glucogallin to di- and trigalloylglucopyranoses and this,
to be emphasized, in the absence of any other cofactors. This unexpected
result could be explained only by the assumption that β-glucogallin had a
dual role in these systems, functioning not only as an acyl acceptor, but
also as an efficient acyl donor to yield a higher galloylated derivative and
free glucose as a by-product (Gross, 1983a). Such a behaviour would
require that β-glucogallin had the unexpected characteristic of an energy-
rich “activated” compound. Considering the known and comparatively
low group-transfer potentials Δ
G
o
' of acylglucoses (
e.g.
, glucose-1-
phosphate,
ca.
21 kJ mol
-1
or glucose-6-phosphate,
ca.
10.5 kJ mol
-1
;
Atkinson and Morton, 1960), the existence of such a reaction was
surprising. The logical classification of β-glucogallin as an activated
compound was later supported by Mock and Strack (1993) that reported
that a related ester, 1-
O
-sinapoyl-β-
D
-glucopyranose, had an
unexpectedly high Δ
G
o
' value of 35.7 kJ mol
-1
. This value is comparable
to that of the well-known acyl-CoA thioesters, and it is reasonable to
assume that the Δ
G
o
' of β-glucogallin is on the same order of magnitude.
Consequently, glucose esters lacking the energy-rich 1-
O
-acyl group
should display no donor function, a conclusion that was corroborated in