Environmental Engineering Reference
In-Depth Information
3
119
Steam
167
0
(d)
(e)
(a)
168
-3
(b)
(c)
131
-6
(f)
(g)
117
-9
-12
169
162
-15
-1
0.78
0.88
0.98
1.08
1.18
1.28
1000/T (K
-1
)
FIGURE 10.5
Arrhenius plot for H
2
O-biochar reaction. (a) Straw, (b) poplar, (c) bark,
(d) beech, (e) birch, (f) maple, and (g) pine. (Source: Reproduced with permission from Di
Blasi (2009). © Elsevier; numbers correspond to references in that publication.)
k
3
CO+ C
f
s
CO
ðÞ!
ðÞ
ð
RX
:
10
:
4c
Þ
In these expressions, C
f
represents an available active carbon site and C(O) an occu-
pied site or, alternatively stated, a carbon
oxygen complex/transitional surface oxide.
CO can have an inhibiting effect on the reaction rate, which consists of lowering the
steady-state concentration of C(O) complexes by increased reverse reaction 1b. The
rate expression is of the Langmuir
-
-
Hinshelwood type:
k
1f
p
CO
2
r
=
ð
Eq
:
10
:
8
Þ
1+
k
1f
k
3
p
CO
2
+
k
1b
k
3
p
CO
Often, a further simplification to n
th
-order kinetics is applied:
r
=k
p
CO
2
ð
Eq
:
10
:
9
Þ
The char gasification reaction with H
2
O is more complex than the char
CO
2
reaction,
because more types of molecules are involved. Basically, there are two models of the
reaction mechanism: the oxygen exchange model and the hydrogen inhibition model,
as summarized by Barrio et al. (2001):
-
+H
2
O
!
k
1b
:
:
C
f
s
ðÞ
CO
ðÞ
+H
2
ð
RX
10
3b
Þ
















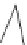




































Search WWH ::

Custom Search