Environmental Engineering Reference
In-Depth Information
mining
Fertilizer
excretion
Guano
agriculture
weathering
uptake by
autotrophs
uptake by
autotrophs
leaching, runoff
Dissolved
in Ocean
Water
Dissolved
in Soil Water,
Lakes, Rivers
Marine
Food
Webs
Land
Food
Webs
death,
decomposition
death,
decomposition
weathering
sedimentation
settling out
uplifting over
geologic time
Rocks
Marine Sediments
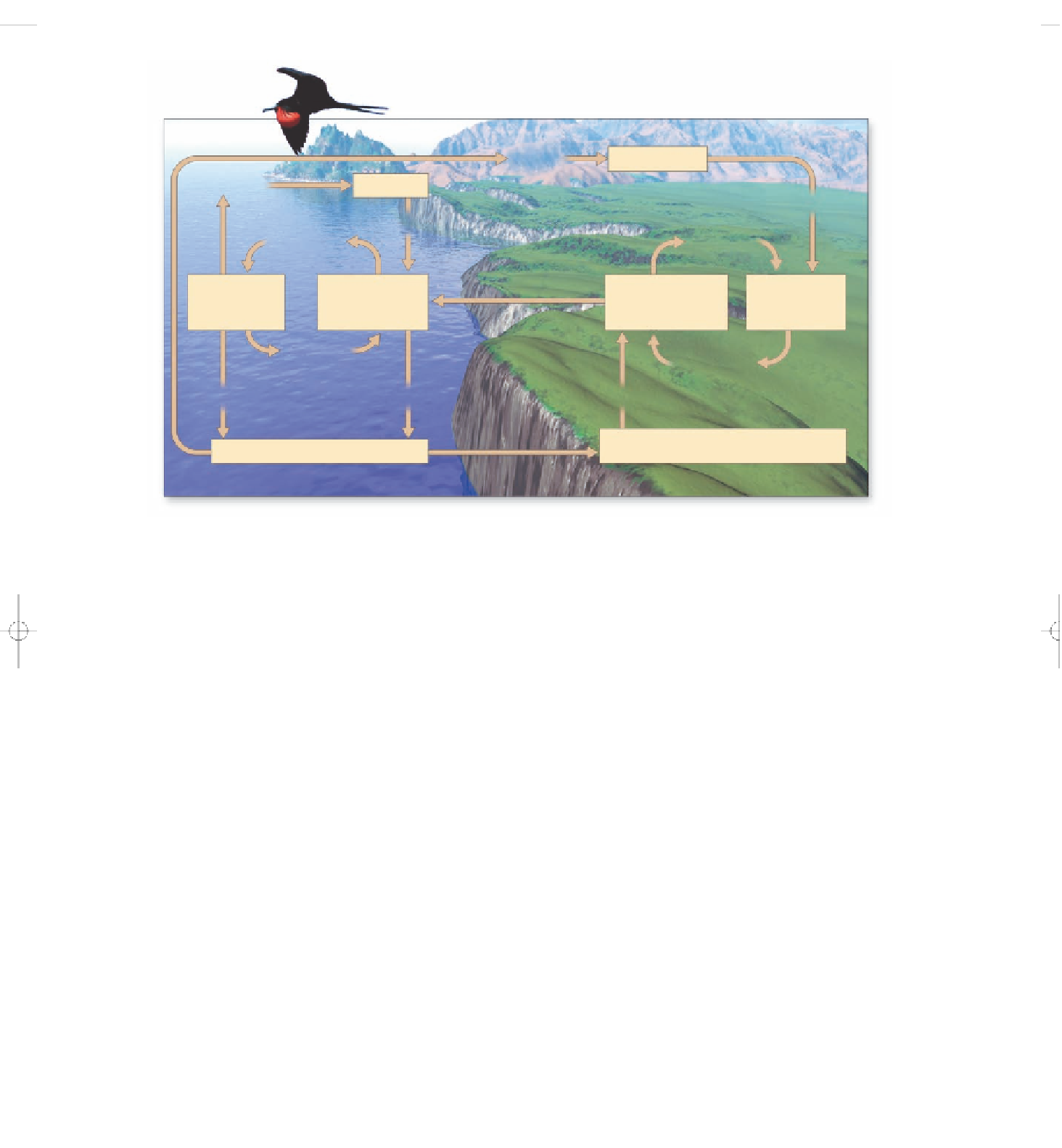
Figure 3-29
Natural capital:
simplified model of the
phosphorus cycle.
Phosphorus reservoirs are shown as
boxes; processes that change one form of phosphorus to another are shown in unboxed print. (From Cecie
Starr and Ralph Taggart,
Biology: The Unity and Diversity of Life,
9th ed., Belmont, Calif.: Wadsworth © 2001)
ions and incorporate the sulfur as an essential compo-
nent of many proteins.
Certain marine algae produce large amounts of
volatile dimethyl sulfide, or DMS (CH
3
SCH
3
). Tiny
droplets of DMS serve as nuclei for the condensation
of water into droplets found in clouds. In this way,
changes in DMS emissions can affect cloud cover and
climate. In the atmosphere, DMS is converted to sulfur
dioxide.
In the atmosphere, sulfur dioxide (SO
2
)fromnat-
ural sources and human activities is converted to sulfur
trioxide gas (SO
3
) and to tiny droplets of sulfuric acid
(H
2
SO
4
). In addition, it reacts with other atmospheric
chemicals such as ammonia to produce tiny particles of
sulfate salts. These droplets and particles fall to the
earth as components of
acid deposition,
which along with
other air pollutants can harm trees and aquatic life.
rock to make commercial inorganic fertilizers and de-
tergents.
Second,
we reduce the available phosphate in
tropical soils when we cut down tropical forests.
Third,
we disrupt aquatic systems with phosphates
from runoff of animal wastes and fertilizers and dis-
charges from sewage treatment systems.
Since 1900, human activities have increased the
natural rate of phosphorus release into the environ-
ment by about 3.7-fold.
The Sulfur Cycle
Sulfur cycles through the earth's air, water, soil, and
living organisms.
Sulfur circulates through the biosphere in the
sulfur
cycle,
shown in Figure 3-30 (p. 60). Much of the earth's
sulfur is stored underground in rocks and minerals, in-
cluding sulfate (SO
4
) salts buried deep under ocean
sediments.
Sulfur also enters the atmosphere from several
natural sources. Hydrogen sulfide (H
2
S)—a colorless,
highly poisonous gas with a rotten-egg smell—is
released from active volcanoes and from organic mat-
ter in flooded swamps, bogs, and tidal flats broken
down by anaerobic decomposers. Sulfur dioxide (SO
2
),
a colorless, suffocating gas, also comes from volcanoes.
Particles of sulfate (SO
4
2
) salts, such as ammo-
nium sulfate, enter the atmosphere from sea spray,
dust storms, and forest fires. Plant roots absorb sulfate
Effects of Human Activities
on the Sulfur Cycle
We add sulfur dioxide to the atmosphere by burning
coal and oil, refining oil, and producing some metals
from ores.
We add sulfur dioxide to the atmosphere in three ways.
First
,weburn sulfur-containing coal and oil to produce
electric power.
Second
,werefine sulfur-containing
petroleum to make gasoline, heating oil, and other
useful products.
Third
,weconvert sulfur-containing