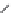Biology Reference
In-Depth Information
rabbit (or human ventricle), and this drives rapid Ca
2
þ
extrusion via Na
þ
/Ca
2
þ
exchange at a time when [Ca
2
þ
]
i
is very high (
Fig. 6
B). In the rabbit ventricle, the
longer action potential plateau keeps Na
þ
/Ca
2
þ
exchange in check, delaying
extrusion until a later time where [Ca
2
þ
]
i
is lower. Another implication of
Fig. 5
B is that there is net Ca
2
þ
e
Z
ux during the contraction in rat (vs. net influx
in rabbit). This means that there must be net Ca
2
þ
influx between contractions in
rat ventricle, and the [Ca
2
þ
]
o
trace in
Fig. 5
B is actually going below the bath by the
end of the trace to restore the steady state balance before the next beat (
1.5 s
later). Note that during a steady state heartbeat, total Ca
2
þ
influx must equal total
Ca
2
þ
e
ux (i.e., there is no net gain or loss of Ca
2
þ
at the steady state).
Extracellular Ca
2
þ
-MEs are also useful for assessing nonsteady state Ca
2
þ
fluxes
on a longer time scale (
Bers and MacLeod, 1986; MacLeod and Bers, 1987
).
Figure 7
A shows that when 0.5 Hz stimulation is stopped there is a very slow
small rise in [Ca
2
þ
]
o
over many seconds (net Ca
2
þ
e
Z
ux), and upon resumption of
stimulation (now at 1 Hz) that there is a net [Ca
2
þ
]
o
depletion which develops over
Z
A
B
30
30
Rat ventricle
Rabbit ventricle
aNa
i
= 7.2 mM
aNa
i
= 7.2 mM
0
−
30
E
Na/Ca
E
Na/Ca
E
m
−
60
60
E
m
−
90
Ca efflux
30
0
−
30
Ca influx
−
60
0
250
500
0
250
500
Time (ms)
Time (ms)
Fig. 6
Changes in the reversal potential of the Na
þ
/Ca
2
þ
exchange (E
Na/Ca
) during the action
potential (E
m
) and Ca
2
þ
transient in rabbit and rat ventricle. Changes in electrochemical driving force
for Na
þ
/Ca
2
þ
exchange (E
Na/Ca
E
m
) are shown in the bottom panels, assuming a 3:1 stoichiometry of
Na
þ
/Ca
2
þ
exchanger and aNa
i
are measured Na
þ
activity values (
Shattock & Bers, 1989
). Ca
2
þ
transients driving the contraction are assumed to be the same for both species (resting [Ca
2
þ
]
i
¼
150 nM,
peak [Ca
2
þ
]
i
¼
1
m
M, 40 ms after the AP initiation). Note that Ca
2
þ
e
Z
ux is low during rest in
rabbit myocytes because of the low [Ca
2
þ
]
i
(despite a significant driving force). Based on data in
Shattock & Bers, (1989)
, as modified in Bers (2001), with permission.