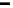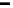Biology Reference
In-Depth Information
D. Correction for Ca
2
þ
Bu
ering
The presence of Ca
2
þ
bu
V
V
ers or binding agents with the appropriate a
Y
nity can
lead to collapse of Ca
2
þ
gradients by shuttle bu
V
ering (
Speksnijder et al., 1989
).
Ca
2
þ
can di
V
use from the surface of the cell in either its free state or bound to the
er. CaSMs only measure the free concentration of Ca
2
þ
. The actual Ca
2
þ
flux at a source is the sum of the measured free Ca
2
þ
flux and the unmeasured
Ca
2
þ
flux di
V
bu
using as Ca
2
þ
bound to bu
V
V
er.
ð
9
Þ
Knowing the conditions under which the Ca
2
þ
flux was measured including the
[Ca
2
þ
] of medium, dissociation constant, K
d
, of the bu
J
Ca total
¼ J
Ca measured
þ J
Ca Buffer
V
ers and concentration of
the bu
ers present, a simple relationship can be derived to determine the ratio of
Ca
2
þ
di
V
using Ca
2
þ
. Shuttle
V
using bound to bu
V
er compared to the freely di
V
ering of H
þ
is a bigger concern than for Ca
2
þ
due to the larger number of
H
þ
bu
V
bu
V
ers that are used in physiological media. The equations that exist to correct
for shuttle bu
ering of H
þ
(
Messerli et al., 2006; Smith et al., 2007
) can be adapted
for use with Ca
2
þ
flux correction and may be necessary under specific
circumstances.
V
D
B
D
Ca
2
þ
K
d
x
i
¼
½
B
ð
10
Þ
2
Ca
2
þ
K
d
þ
The correction factor, ''x
i
'', is the ratio of the Ca
2
þ
bound bu
V
er flux to the free
Ca
2
þ
flux. Therefore
J
Ca total
¼ J
Ca measured
1
ð
þ x
i
þþx
n
Þ
ð
11
Þ
ers (x
i
þþ
x
n
) may be collapsing the Ca
2
þ
gradient. The correction factor is based on three criteria, the ratio of the di
erent Ca
2
þ
bu
where a number of di
V
V
V
usion
cients of the Ca
2
þ
-bu
er complex to free Ca
2
þ
,theCa
2
þ
bu
coe
Y
V
V
er concentration,
and the dissociation constant, K
d
,oftheCa
2
þ
bu
er compared to the [Ca
2
þ
]ofthe
medium. The K
d
is the inverse of the more commonly used ''stability constant'' also
known as the association constant. Only Ca
2
þ
bu
V
ers/binding agents that have K
d
values near the range of the extracellular [Ca
2
þ
] will act as e
V
ers
during self-referencing of CaSMs.
Table III
lists a few of these compounds. Note
that two of the compounds, ADA and Bicine, are commonly used as H
þ
bu
V
ective shuttle bu
V
V
ers.
Generally, the Ca
2
þ
K
d
values of other Good bu
ers are not in the range of normal
extracellular [Ca
2
þ
] or are very poor Ca
2
þ
chelators (
Dawson et al., 1986
).
V
E. Measurement of Voltage Gradients
The use of CaSMs with self-referencing is subject to a similar problem that
occurs with the use of intracellular CaSMs; specifically they detect not only
changes in ion concentration but also voltage. Extracellular voltage gradients