Chemistry Reference
In-Depth Information
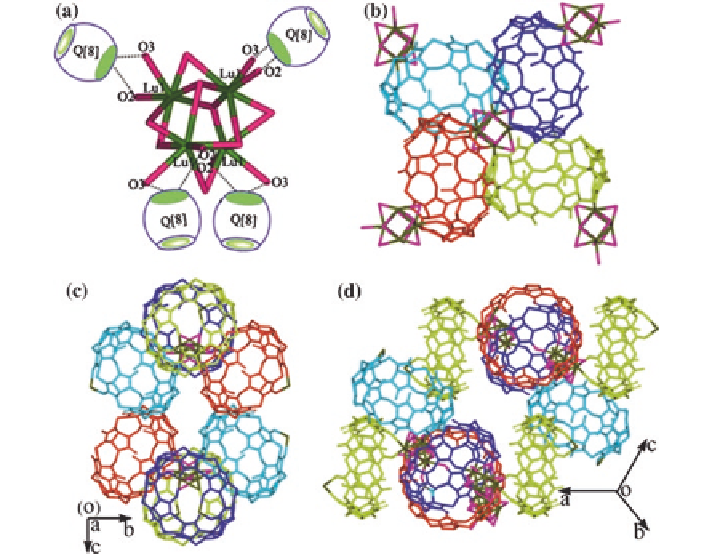
Fig. 4.9 a
A Lu
3
+
-unit;
b
a basic four-Q[8] building block;
c
-
d
3D frameworks based on the
direct coordination of Lu
3
+
cations to Q[8] molecules along different directions [
13
]
surrounded by six [CdCl
4
]
2
−
anions through the so-called outer-surface inter-
action of Q[
n
]s. The coordination of Lu
3
+
with Q[8] molecules in the presence
of [CdCl
4
]
2
−
anions served as a representative isomorphous complex of the
Ln
3
heavy
−
Q[8]
−
[CdCl
4
]
2
−
group, showed an unexpected experimental result. An
Lu
3
+
-unit containing four Lu
3
+
cations with a 50 % occupancy links four Q[8]
molecules as a basic building block (Fig.
4.9
a, b) and each Lu
3
+
-unit links four
Q[8] molecules and forms the basic four-Q[8] building block. In turn, each Q[8]
molecule in the block coordinates with two Lu
3
+
-units. Moreover, the extra hydro-
gen bonding of
≡
CH or
=
CH
2
groups on the back of a Q[8] molecule with portal
carbonyl oxygens of Q[8] exist between every two adjacent Q[8] molecules in the
block (Fig.
4.9
b). Thus, extending the basic building block results in the formation
of a novel 3D framework with various channels, through the direct coordination of
Lu
3
+
cations to Q[8] molecules (Fig.
4.9
c, d).
Most interestingly, systems containing the light lanthanides and Q[8] in 6 mol
aqueous HCl have been found to give crystals more rapidly than systems con-
taining the heavy lanthanides. This suggests that the difference could be used to
separate the lighter lanthanides cations from their heavier counterparts. When a
solution of Q[8] in aqueous HCl was added to mixtures of light and heavy lan-
thanides in a 1:1 ratio, for example Ce:Lu, in aqueous HCl solution, stick-like
Search WWH ::

Custom Search